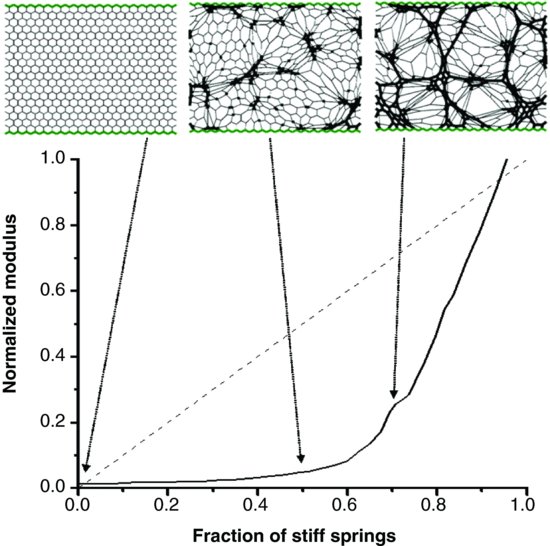
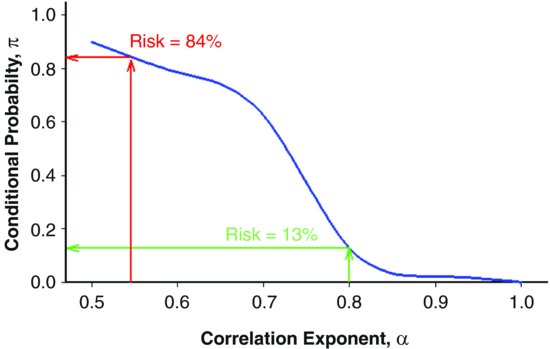
| References |
| 1. |
Akay M,
Moodie KL,
Hoopes PJ.
Age related alterations in the complexity of respiratory patterns.
J Integr Neurosci
2:
165-178,
2003.
|
| 2. |
Akay M,
Mulder EJ.
Effects of maternal alcohol intake on fractal properties in human fetal breathing dynamics.
IEEE Trans Biomed Eng
45:
1097-1103,
1998.
|
| 3. |
Akay M,
Sekine N.
Investigating the complexity of respiratory patterns during recovery from severe hypoxia.
J Neural Eng
1:
16-20,
2004.
|
| 4. |
Albert R,
Jeong H, and
Barabasi AL.
Error and attack tolerance of complex networks.
Nature
406:
378-382,
2000.
|
| 5. |
Alencar AM,
Arold SP,
Buldyrev SV,
Majumdar A,
Stamenovic D,
Stanley HE,
Suki B.
Physiology: Dynamic instabilities in the inflating lung.
Nature
417:
809-811,
2002.
|
| 6. |
Alencar AM,
Buldyrev SV,
Majumdar A,
Stanley HE,
Suki B.
Avalanche dynamics of crackle sound in the lung.
Phys Rev Lett
87:
088101,
2001.
|
| 7. |
Alencar AM,
Hantos Z,
Petak F,
Tolnai J,
Asztalos T,
Zapperi S,
Andrade JS Jr,
Buldyrev SV,
Stanley HE,
Suki B.
Scaling behavior in crackle sound during lung inflation.
Phys Rev E Stat Phys Plasmas Fluids Relat Interdiscip Topics
60:
4659-4663,
1999.
|
| 8. |
Amin SD,
Majumdar A,
Frey U,
Suki B.
Modeling the dynamics of airway constriction: Effects of agonist transport and binding.
J Appl Physiol
109:
553-563,
2010.
|
| 9. |
Aon MA,
Cortassa S,
O'Rourke B.
Percolation and criticality in a mitochondrial network.
Proc Natl Acad Sci U S A
101:
4447-4452,
2004.
|
| 10. |
Arold SP,
Bartolak‐Suki E,
Suki B.
Variable stretch pattern enhances surfactant secretion in alveolar type II cells in culture.
Am J Physiol Lung Cell Mol Physiol
296:
L574-581,
2009.
|
| 11. |
Arold SP,
Mora R,
Lutchen KR,
Ingenito EP,
Suki B.
Variable tidal volume ventilation improves lung mechanics and gas exchange in a rodent model of acute lung injury.
Am J Respir Crit Care Med
165:
366-371,
2002.
|
| 12. |
Arold SP,
Suki B,
Alencar AM,
Lutchen KR,
Ingenito EP.
Variable ventilation induces endogenous surfactant release in normal guinea pigs.
Am J Physiol Lung Cell Mol Physiol
285:
L370-375,
2003.
|
| 13. |
Bak P.
How nature Works: The Science of Self‐Organized Criticality.
New York, NY, USA:
Copernicus,
1996.
|
| 14. |
Baldwin DN,
Suki B,
Pillow JJ,
Roiha HL,
Minocchieri S,
Frey U.
Effect of sighs on breathing memory and dynamics in healthy infants.
J Appl Physiol
97:
1830-1839,
2004.
|
| 15. |
Banavar JR,
Maritan A,
Rinaldo A.
Size and form in efficient transportation networks.
Nature
399:
130-132,
1999.
|
| 16. |
Barabási A‐L,
Stanley HE.
Fractal Concepts in Surface Growth.
New York, NY:
Press Syndicate of the University of Cambridge,
1995.
|
| 17. |
Barabasi AL,
Buldyrev SV,
Stanley HE,
Suki B.
Avalanches in the lung: A statistical mechanical model.
Phys Rev Lett
76:
2192-2195,
1996.
|
| 18. |
Barnes PJ.
Chronic obstructive pulmonary disease.
N Engl J Med
343:
269-280,
2000.
|
| 19. |
Barnes PJ,
Stockley RA.
COPD: Current therapeutic interventions and future approaches.
Eur Respir J
25:
1084-1106,
2005.
|
| 20. |
Bassingthwaighte JB,
Raymond GM.
Evaluating rescaled ranged analysis for time series.
Ann Biomed Eng
22:
432-444,
1994.
|
| 21. |
Bates JH.
A recruitment model of quasi‐linear power‐law stress adaptation in lung tissue.
Ann Biomed Eng
35:
1165-1174,
2007.
|
| 22. |
Bates JH,
Davis GS,
Majumdar A,
Butnor KJ,
Suki B.
Linking parenchymal disease progression to changes in lung mechanical function by percolation.
Am J Respir Crit Care Med
176:
617-623,
2007.
|
| 23. |
Bates JHT.
Nonlinear network theory of complex disease. In
Bar‐Yam Y, editor.
International Conference on Complex Systems.
Boston, MA.
New England Complex Systems Institute,
2006, p.
28.
|
| 24. |
Bellardine CL,
Hoffman AM,
Tsai L,
Ingenito EP,
Arold SP,
Lutchen KR,
Suki B.
Comparison of variable and conventional ventilation in a sheep saline lavage lung injury model.
Crit Care Med
34:
439-445,
2006.
|
| 25. |
Berisio R,
Granata V,
Vitagliano L,
Zagari A.
Imino acids and collagen triple helix stability: Characterization of collagen‐like polypeptides containing Hyp‐Hyp‐Gly sequence repeats.
J Am Chem Soc
126:
11402-11403,
2004.
|
| 26. |
Black LD,
Allen PG,
Morris SM,
Stone PJ,
Suki B.
Mechanical and failure properties of extracellular matrix sheets as a function of structural protein composition.
Biophys J
94:
1916-1929,
2008.
|
| 27. |
Boker A,
Haberman CJ,
Girling L,
Guzman RP,
Louridas G,
Tanner JR,
Cheang M,
Maycher BW,
Bell DD,
Doak GJ.
Variable ventilation improves perioperative lung function in patients undergoing abdominal aortic aneurysmectomy.
Anesthesiology
100:
608-616,
2004.
|
| 28. |
Bonnet R,
Jorres R,
Heitmann U,
Magnussen H.
Circadian rhythm in airway responsiveness and airway tone in patients with mild asthma.
J Appl Physiol
71:
1598-1605,
1991.
|
| 29. |
Boser SR,
Park H,
Perry SF,
Menache MG,
Green FH.
Fractal geometry of airway remodeling in human asthma.
Am J Respir Crit Care Med
172:
817-823,
2005.
|
| 30. |
Botros SM,
Bruce EN.
Neural network implementation of a three‐phase model of respiratory rhythm generation.
Biol Cybern
63:
143-153,
1990.
|
| 31. |
Bray D.
Molecular networks: The top‐down view.
Science
301:
1864-1865,
2003.
|
| 32. |
Brenner M,
McKenna RJ Jr,
Gelb AF,
Fischel RJ,
Wilson AF.
Rate of FEV1 change following lung volume reduction surgery.
Chest
113:
652-659,
1998.
|
| 33. |
Bruce EN.
Temporal variations in the pattern of breathing.
J Appl Physiol
80:
1079-1087,
1996.
|
| 34. |
Buard B,
Mahe G,
Chapeau‐Blondeau F,
Rousseau D,
Abraham P,
Humeau A.
Generalized fractal dimensions of laser Doppler flowmetry signals recorded from glabrous and nonglabrous skin.
Med Phys
37:
2827-2836,
2010.
|
| 35. |
Buldyrev SV.
Fractals in biology, In: Encyclopedia of Complexity and Systems Science, edited by Meyers RA. New York: Springer,
2009,
p. 3779-‐3802
|
| 36. |
Bunde A,
Havlin S.
Fractals and Disordered Systems.
Berlin, NY:
Springer,
1996.
|
| 37. |
Burioka N,
Miyata M,
Endo M,
Fukuoka Y,
Suyama H,
Nakazaki H,
Igawa K,
Shimizu E.
Alteration of the circadian rhythm in peak expiratory flow of nocturnal asthma following nighttime transdermal beta2‐adrenoceptor agonist tulobuterol chronotherapy.
Chronobiol Int
22:
383-390,
2005.
|
| 38. |
Butler JP,
Tsuda A.
Comment on “interplay between geometry and flow distribution in an airway tree”.
Phys Rev Lett
93:
049801; author reply 049802,
2004.
|
| 39. |
Caldirola D,
Bellodi L,
Caumo A,
Migliarese G,
Perna G.
Approximate entropy of respiratory patterns in panic disorder.
Am J Psychiatry
161:
79-87,
2004.
|
| 40. |
Cates ME.
Reptation of living polymers: Dynamics of entangled polymers in the presence of reversible chain‐scission reactions.
Macromolecules
20:
2289-2296,
1987.
|
| 41. |
Cavalcante FS,
Ito S,
Brewer KK,
Sakai H,
Alencar AM,
Almeida MP,
Andrade JS Jr,
Majumdar A,
Ingenito EP,
Suki B.
Mechanical interactions between collagen and proteoglycans: Implications for the stability of lung tissue.
J Appl Physiol
98:
672-679,
2005.
|
| 42. |
Cernelc M,
Suki B,
Reinmann B,
Hall GL,
Frey U.
Correlation properties of tidal volume and end‐tidal O2 and CO2 concentrations in healthy infants.
J Appl Physiol
92:
1817-1827,
2002.
|
| 43. |
Collard HR,
Moore BB,
Flaherty KR,
Brown KK,
Kaner RJ,
King TE Jr,
Lasky JA,
Loyd JE,
Noth I,
Olman MA,
Raghu G,
Roman J,
Ryu JH,
Zisman DA,
Hunninghake GW,
Colby TV,
Egan JJ,
Hansell DM,
Johkoh T,
Kaminski N,
Kim DS,
Kondoh Y,
Lynch DA,
Muller‐Quernheim J,
Myers JL,
Nicholson AG,
Selman M,
Toews GB,
Wells AU,
Martinez FJ.
Acute exacerbations of idiopathic pulmonary fibrosis.
Am J Respir Crit Care Med
176:
636-643,
2007.
|
| 44. |
Coughlin MF,
Suki B,
Stamenovic D.
Dynamic behavior of lung parenchyma in shear.
J Appl Physiol
80:
1880-1890,
1996.
|
| 45. |
Coveney PV,
Fowler PW.
Modelling biological complexity: A physical scientist's perspective.
J R Soc Interface
2:
267-280,
2005.
|
| 46. |
Crawford AB,
Cotton DJ,
Paiva M,
Engel LA.
Effect of airway closure on ventilation distribution.
J Appl Physiol
66:
2511-2515,
1989.
|
| 47. |
D'Alonzo GE,
Steinijans VW,
Keller A.
Measurements of morning and evening airflow grossly underestimate the circadian variability of FEV1 and peak expiratory flow rate in asthma.
Am J Respir Crit Care Med
152:
1097-1099,
1995.
|
| 48. |
Dawson CD,
Krenz GS,
Lineha JH.
Complexity and structure‐function relationships in the pulmonary arterial tree. In:
Hlastala M.P,
Robertson HT, editors.
Lung Biology in Health and Disease, Complexity in Structure and Function of the Lung.
New York:
Marcel Dekker, Inc.,
1998, p.
401-427.
|
| 49. |
de Gennes PG.
Reptation of a polymer chain in the presence of fixed obstacles.
J Chem Phys
55:
572-579,
1971.
|
| 50. |
Dewey TG.
Fractals in Molecular Biophysics.
Oxford, NY:
Oxford University Press,
1997.
|
| 51. |
Diba C,
Salome CM,
Reddel HK,
Thorpe CW,
Toelle B,
King GG.
Short‐term variability of airway caliber‐a marker of asthma?
J Appl Physiol
103:
296-304,
2007.
|
| 52. |
Ding DJ,
Martin JG,
Macklem PT.
Effects of lung volume on maximal methacholine‐induced bronchoconstriction in normal humans.
J Appl Physiol
62:
1324-1330,
1987.
|
| 53. |
Dirksen A,
Holstein‐Rathlou NH,
Madsen F,
Skovgaard LT,
Ulrik CS,
Heckscher T,
Kok‐Jensen A.
Long‐range correlations of serial FEV1 measurements in emphysematous patients and normal subjects.
J Appl Physiol
85:
259-265,
1998.
|
| 54. |
Doi M,
Edwards SF.
The Theory of Polymer Dynamics.
New York:
Oxford University Press,
1986.
|
| 55. |
Dreher D,
Koller EA.
Circadian rhythms of specific airway conductance and bronchial reactivity to histamine: The effects of parasympathetic blockade.
Eur Respir J
3:
414-420,
1990.
|
| 56. |
Ebihara T,
Venkatesan N,
Tanaka R,
Ludwig MS.
Changes in extracellular matrix and tissue viscoelasticity in bleomycin‐induced lung fibrosis. Temporal aspects.
Am J Respir Crit Care Med
162:
1569-1576,
2000.
|
| 57. |
Edelman GM.
Topobiology : An Introduction to Molecular Embryology.
New York:
Basic Books,
1988.
|
| 58. |
Engler AJ,
Humbert PO,
Wehrle‐Haller B,
Weaver VM.
Multiscale modeling of form and function.
Science
324:
208-212,
2009.
|
| 59. |
Engler AJ,
Sen S,
Sweeney HL,
Discher DE.
Matrix elasticity directs stem cell lineage specification.
Cell
126:
677-689,
2006.
|
| 60. |
Engoren M.
Approximate entropy of respiratory rate and tidal volume during weaning from mechanical ventilation.
Crit Care Med
26:
1817-1823,
1998.
|
| 61. |
Engoren M,
Courtney SE,
Habib RH.
Effect of weight and age on respiratory complexity in premature neonates.
J Appl Physiol
106:
766-773,
2009.
|
| 62. |
Fabry B,
Maksym GN,
Butler JP,
Glogauer M,
Navajas D,
Fredberg JJ.
Scaling the microrheology of living cells.
Phys Rev Lett
87:
148102,
2001.
|
| 63. |
Fadel PJ,
Barman SM,
Phillips SW,
Gebber GL.
Fractal fluctuations in human respiration.
J Appl Physiol
97:
2056-2064,
2004.
|
| 64. |
Farber JP.
Medullary inspiratory activity during opossum development.
Am J Physiol
254:
R578-584,
1988.
|
| 65. |
Felici M,
Filoche M,
Straus C,
Similowski T,
Sapoval B.
Diffusional screening in real 3D human acini—a theoretical study.
Respir Physiol Neurobiol
145:
279-293,
2005.
|
| 66. |
Ferry J.
Viscoelastic Properties of Polymers.
New York:
Wiley,
1980.
|
| 67. |
Fetter LJ,
Kiss AD,
Pearson, DS,
Quack GS,
Vitus FJ.
Rheological behavior of star‐shaped polymers.
Macromolecules
26:
647-654,
1993.
|
| 68. |
Fiamma MN,
Samara Z,
Baconnier P,
Similowski T,
Straus C.
Respiratory inductive plethysmography to assess respiratory variability and complexity in humans.
Respir Physiol Neurobiol
156:
234-239,
2007.
|
| 69. |
Florens M,
Sapoval B,
Filoche M.
The optimal branching asymmetry of a bidirectional distribution tree.
Computer Physics Communications,
2010, doi:10.1016/j.cpc.2010.11.008.
|
| 70. |
Forgacs G.
On the possible role of cytoskeletal filamentous networks in intracellular signaling: An approach based on percolation.
J Cell Sci
108:
(Pt 6):
2131-2143,
1995.
|
| 71. |
Fratzl P,
Daxer A.
Structural transformation of collagen fibrils in corneal stroma during drying. x‐ray scattering study An.
Biophys J
64:
1210-1214,
1993.
|
| 72. |
Fredberg JJ,
Inouye D,
Miller B,
Nathan M,
Jafari S,
Raboudi SH,
Butler JP,
Shore SA.
Airway smooth muscle, tidal stretches, and dynamically determined contractile states.
Am J Respir Crit Care Med
156:
1752-1759,
1997.
|
| 73. |
Fredberg JJ,
Jones KA,
Nathan M,
Raboudi S,
Prakash YS,
Shore SA,
Butler JP,
Sieck GC.
Friction in airway smooth muscle: Mechanism, latch, and implications in asthma.
J Appl Physiol
81:
2703-2712,
1996.
|
| 74. |
Fredberg JJ,
Kamm RD.
Stress transmission in the lung: Pathways from organ to molecule.
Annu Rev Physiol
68:
507-541,
2006.
|
| 75. |
Frey U,
Brodbeck T,
Majumdar A,
Taylor DR,
Town GI,
Silverman M,
Suki B.
Risk of severe asthma episodes predicted from fluctuation analysis of airway function.
Nature
438:
667-670,
2005.
|
| 76. |
Frey U,
Hislop A,
Silverman M.
Branching properties of the pulmonary arterial tree during pre‐ and postnatal development.
Respir Physiol Neurobiol
139:
179-189,
2004.
|
| 77. |
Frey U,
Silverman M,
Barabasi AL,
Suki B.
Irregularities and power law distributions in the breathing pattern in preterm and term infants.
J Appl Physiol
85:
789-797,
1998.
|
| 78. |
Frey U,
Suki B.
Complexity of chronic asthma and chronic obstructive pulmonary disease: Implications for risk assessment, and disease progression and control.
Lancet
372:
1088-1099,
2008.
|
| 79. |
Fung YC.
Biomechanics: Mechanical Properties of Living Tissues.
New York:
Springer‐Verlag,
1993.
|
| 80. |
Gelb AF,
McKenna RJ Jr,
Brenner M,
Schein MJ,
Zamel N,
Fischel R.
Lung function 4 years after lung volume reduction surgery for emphysema.
Chest
116:
1608-1615,
1999.
|
| 81. |
Gillis HL,
Lutchen KR.
How heterogeneous bronchoconstriction affects ventilation distribution in human lungs: A morphometric model.
Ann Biomed Eng
27:
14-22,
1999.
|
| 82. |
Glenny R,
Bernard S,
Neradilek B,
Polissar N.
Quantifying the genetic influence on mammalian vascular tree structure.
Proc Natl Acad Sci U S A
104:
6858-6863,
2007.
|
| 83. |
Glenny RW.
Spatial correlation of regional pulmonary perfusion.
J Appl Physiol
72:
2378-2386,
1992.
|
| 84. |
Glenny RW,
Robertson HT.
A computer simulation of pulmonary perfusion in three dimensions.
J Appl Physiol
79:
357-369,
1995.
|
| 85. |
Glenny RW,
Robertson HT.
Fractal modeling of pulmonary blood flow heterogeneity.
J Appl Physiol
70:
1024-1030,
1991.
|
| 86. |
Glenny RW,
Robertson HT.
Fractal properties of pulmonary blood flow: Characterization of spatial heterogeneity.
J Appl Physiol
69:
532-545,
1990.
|
| 87. |
Glenny RW,
Robertson HT,
Hlastala MP.
Vasomotor tone does not affect perfusion heterogeneity and gas exchange in normal primate lungs during normoxia.
J Appl Physiol
89:
2263-2267,
2000.
|
| 88. |
Glenny RW,
Robertson HT,
Yamashiro S,
Bassingthwaighte JB.
Applications of fractal analysis to physiology.
J Appl Physiol
70:
2351-2367,
1991.
|
| 89. |
Goldberger AL,
Amaral LA,
Hausdorff JM,
Ivanov P,
Peng CK,
Stanley HE.
Fractal dynamics in physiology: Alterations with disease and aging.
Proc Natl Acad Sci U S A
99:
(Suppl 1):
2466-2472,
2002.
|
| 90. |
Goldstein RH,
Lucey EC,
Franzblau C,
Snider GL.
Failure of mechanical properties to parallel changes in lung connective tissue composition in bleomycin‐induced pulmonary fibrosis in hamsters.
Am Rev Respir Dis
120:
67-73,
1979.
|
| 91. |
Grimmett GR,
Stirzaker DR.
Probability and Random Processes.
New York:
Oxford University Press,
1992.
|
| 92. |
Hantos Z,
Daroczy B,
Suki B,
Nagy S,
Fredberg JJ.
Input impedance and peripheral inhomogeneity of dog lungs.
J Appl Physiol
72:
168-178,
1992.
|
| 93. |
Hantos Z,
Tolnai J,
Asztalos T,
Petak F,
Adamicza A,
Alencar AM,
Majumdar A,
Suki B.
Acoustic evidence of airway opening during recruitment in excised dog lungs.
J Appl Physiol
97:
592-598,
2004.
|
| 94. |
Hart N,
Polkey MI,
Clement A,
Boule M,
Moxham J,
Lofaso F,
Fauroux B.
Changes in pulmonary mechanics with increasing disease severity in children and young adults with cystic fibrosis.
Am J Respir Crit Care Med
166:
61-66,
2002.
|
| 95. |
Hayhurst MD,
MacNee W,
Flenley DC,
Wright D,
McLean A,
Lamb D,
Wightman AJ,
Best J.
Diagnosis of pulmonary emphysema by computerised tomography.
Lancet
2:
320-322,
1984.
|
| 96. |
Haykin SS.
Neural Networks and Learning Machines.
New York:
Prentice Hall,
2009.
|
| 97. |
Hetzel MR,
Clark TJ.
Comparison of normal and asthmatic circadian rhythms in peak expiratory flow rate.
Thorax
35:
732-738,
1980.
|
| 98. |
Hoop B,
Burton MD,
Kazemi H,
Liebovitch LS.
Correlation in stimulated respiratory neural noise.
Chaos
5:
609-612,
1995.
|
| 99. |
Horsfield K,
Dart G,
Olson DE,
Filley GF,
Cumming G.
Models of the human bronchial tree.
J Appl Physiol
31:
207-217,
1971.
|
| 100. |
Hukins DWL.
Connective Tissue Matrix.
London:
Macmillan,
1984.
|
| 101. |
Irvin CG.
Lung volume: A principle determinant of airway smooth muscle function.
Eur Respir J
22:
3-5,
2003.
|
| 102. |
Ito S,
Majumdar A,
Kume H,
Shimokata K,
Naruse K,
Lutchen KR,
Stamenovic D,
Suki B.
Viscoelastic and dynamic nonlinear properties of airway smooth muscle tissue: Roles of mechanical force and the cytoskeleton.
Am J Physiol Lung Cell Mol Physiol
290:
L1227-L1237,
2006.
|
| 103. |
Ivanov PC,
Amaral LA,
Goldberger AL,
Havlin S,
Rosenblum MG,
Struzik ZR,
Stanley HE.
Multifractality in human heartbeat dynamics.
Nature
399:
461-465,
1999.
|
| 104. |
Ivanov PC,
Nunes Amaral LA,
Goldberger AL,
Havlin S,
Rosenblum MG,
Stanley HE,
Struzik ZR.
From 1/f noise to multifractal cascades in heartbeat dynamics.
Chaos
11:
641-652,
2001.
|
| 105. |
Kamm RD.
Airway wall mechanics.
Annu Rev Biomed Eng
1:
47-72,
1999.
|
| 106. |
Kang H,
Koh YY,
Yoo Y,
Yu J,
Kim DK,
Kim CK.
Maximal airway response to methacholine in cough‐variant asthma: Comparison with classic asthma and its relationship to peak expiratory flow variability.
Chest
128:
3881-3887,
2005.
|
| 107. |
Kauffman SA.
At Home in the Universe: The Search for Laws of Self‐Organization and Complexity.
New York:
Oxford University Press,
1995.
|
| 108. |
Kerr HD.
Diurnal variation of respiratory function independent of air quality: Experience with an environmentally controlled exposure chamber for human subjects.
Arch Environ Health
26:
144-152,
1973.
|
| 109. |
Kirkpatrick S,
Gelatt CD,
Vecchi MP Jr.
Optimization by simulated annealing.
Science
220:
671-680,
1983.
|
| 110. |
Kitaoka H,
Suki B.
Branching design of the bronchial tree based on a diameter‐flow relationship.
J Appl Physiol
82:
968-976,
1997.
|
| 111. |
Kitaoka H,
Takaki R,
Suki B.
A three‐dimensional model of the human airway tree.
J Appl Physiol
87:
2207-2217,
1999.
|
| 112. |
Kleiber M.
Body size and metabolism.
Hilgardia
6:
315-353,
1932.
|
| 113. |
Koh YY,
Kang H,
Yoo Y,
Yu J,
Nah KM,
Kim CK.
Peak expiratory flow variability and exercise responsiveness in methacholine‐hyperresponsive adolescents with asthma remission.
J Asthma
42:
17-23,
2005.
|
| 114. |
Kolokotrones T,
Van S,
Deeds EJ,
Fontana W.
Curvature in metabolic scaling.
Nature
464:
753-756,
2010.
|
| 115. |
Kononov S,
Brewer K,
Sakai H,
Cavalcante FS,
Sabayanagam CR,
Ingenito EP,
Suki B.
Roles of mechanical forces and collagen failure in the development of elastase‐induced emphysema.
Am J Respir Crit Care Med
164:
1920-1926.,
2001.
|
| 116. |
Kronick PL,
Sacks MS.
Matrix macromolecules that affect the viscoelasticity of calfskin.
J Biomech Eng
116:
140-145,
1994.
|
| 117. |
Lall CA,
Cheng N,
Hernandez P,
Pianosi PT,
Dali Z,
Abouzied A,
Maksym GN.
Airway resistance variability and response to bronchodilator in children with asthma.
Eur Respir J
30:
260-268,
2007.
|
| 118. |
Larsen PD,
Elder DE,
Tzeng YC,
Campbell AJ,
Galletly DC.
Fractal characteristics of breath to breath timing in sleeping infants.
Respir Physiol Neurobiol
139:
263-270,
2004.
|
| 119. |
Lefevre GR,
Kowalski SE,
Girling LG,
Thiessen DB,
Mutch WA.
Improved arterial oxygenation after oleic acid lung injury in the pig using a computer‐controlled mechanical ventilator.
Am J Respir Crit Care Med
154:
1567-1572,
1996.
|
| 120. |
Lefevre J.
Teleonomical optimization of a fractal model of the pulmonary arterial bed.
J Theor Biol
102:
225-248,
1983.
|
| 121. |
Liebovitch LS,
Fischbarg J,
Koniarek JP.
Ion channel kinetics: A model based on fractal scaling rather than multistate Markov processes.
Math Biosci
84:
37-68,
1987.
|
| 122. |
Lum H,
Mitzner W.A
species comparison of alveolar size and surface forces.
J Appl Physiol
62:
1865-1871,
1987.
|
| 123. |
Macklem PT.
Can airway function be predicted?
Am J Respir Crit Care Med
153:
S19-20,
1996.
|
| 124. |
Macklem PT,
Seely, A.
Towards a definition of life.
Perspect Biol Med
53(3):
330-‐340,
2010.
|
| 125. |
Majumdar A,
Alencar AM,
Buldyrev SV,
Hantos Z,
Lutchen KR,
Stanley HE,
Suki B.
Relating airway diameter distributions to regular branching asymmetry in the lung.
Phys Rev Lett
95:
168101,
2005.
|
| 126. |
Majumdar A,
Hantos Z,
Tolnai J,
Parameswaran H,
Tepper R,
Suki B.
Estimating the diameter of airways susceptible for collapse using crackle sound.
J Appl Physiol
107:
1504-1512,
2009.
|
| 127. |
Maksym GN,
Bates JH.
A distributed nonlinear model of lung tissue elasticity.
J Appl Physiol
82:
32-41,
1997.
|
| 128. |
Maksym GN,
Fredberg JJ,
Bates JH.
Force heterogeneity in a two‐dimensional network model of lung tissue elasticity.
J Appl Physiol
85:
1223-1229,
1998.
|
| 129. |
Mandelbrot BB.
The Fractal Geometry of Nature.
San Francisco:
W.H. Freeman,
1983.
|
| 130. |
Matsuoka S,
Kurihara Y,
Yagihashi K,
Nakajima Y.
Morphological progression of emphysema on thin‐section CT: Analysis of longitudinal change in the number and size of low‐attenuation clusters.
J Comput Assist Tomogr
30:
669-674,
2006.
|
| 131. |
Mauroy B,
Filoche M,
Weibel ER,
Sapoval B.
An optimal bronchial tree may be dangerous.
Nature
427:
633-636,
2004.
|
| 132. |
McCullagh CM,
Jamieson AM,
Blackwell J,
Gupta R.
Viscoelastic properties of human tracheobronchial mucin in aqueous solution.
Biopolymers
35:
149-159,
1995.
|
| 133. |
McDonnell MD,
Abbott D.
What is stochastic resonance? Definitions, misconceptions, debates, and its relevance to biology.
PLoS Comput Biol
5:
e1000348,
2009.
|
| 134. |
McNamee JE.
Fractal perspectives in pulmonary physiology.
J Appl Physiol
71:
1-8,
1991.
|
| 135. |
Meyers LA,
Newman ME,
Pourbohloul B.
Predicting epidemics on directed contact networks.
J Theor Biol
240:
400-418,
2006.
|
| 136. |
Mijailovich SM,
Butler JP,
Fredberg JJ.
Perturbed equilibria of myosin binding in airway smooth muscle: Bond‐length distributions, mechanics, and ATP metabolism.
Biophys J
79:
2667-2681,
2000.
|
| 137. |
Mijailovich SM,
Stamenovic D,
Fredberg JJ.
Toward a kinetic theory of connective tissue micromechanics.
J Appl Physiol
74:
665-681,
1993.
|
| 138. |
Mishima M,
Hirai T,
Itoh H,
Nakano Y,
Sakai H,
Muro S,
Nishimura K,
Oku Y,
Chin K,
Ohi M,
Nakamura T,
Bates JH,
Alencar AM, and
Suki B.
Complexity of terminal airspace geometry assessed by lung computed tomography in normal subjects and patients with chronic obstructive pulmonary disease.
Proc Natl Acad Sci U S A
96:
8829-8834,
1999.
|
| 139. |
Mortola JP.
Breathing around the clock: An overview of the circadian pattern of respiration.
Eur J Appl Physiol
91:
119-129,
2004.
|
| 140. |
Murray CD.
The physiological principle of minimum work: I. The vascular system and the cost of blood volume.
Proc Natl Acad Sci U S A
12:
207-214,
1926.
|
| 141. |
Muskulus M,
Slats AM,
Sterk PJ,
Verduyn‐Lunel S.
Fluctuations and determinism of respiratory impedance in asthma and chronic obstructive pulmonary disease.
J Appl Physiol
109:
1582-1591,
2010.
|
| 142. |
Nelson TR,
Manchester DK.
Modeling of lung morphogenesis using fractal geometries.
IEEE Trans Med Imaging
7:
321-327,
1988.
|
| 143. |
Nichols JE,
Cortiella J.
Engineering of a complex organ: Progress toward development of a tissue‐engineered lung.
Proc Am Thorac Soc
5:
723-730,
2008.
|
| 144. |
Nicolis G,
Proigogine I.
Exploring Complexity: An Introduction.
New York:
W.H. Freeman & Company,
1989.
|
| 145. |
Oosterhoff Y,
Koeter GH,
De Monchy JG,
Postma DS.
Circadian variation in airway responsiveness to methacholine, propranolol, and AMP in atopic asthmatic subjects.
Am Rev Respir Dis
147:
512-517,
1993.
|
| 146. |
Painter PR,
Eden P,
Bengtsson HU.
Pulsatile blood flow, shear force, energy dissipation and Murray's Law.
Theor Biol Med Model
3:
31,
2006.
|
| 147. |
Parameswaran H,
Majumdar A,
Ito S,
Alencar AM,
Suki B.
Quantitative characterization of airspace enlargement in emphysema.
J Appl Physiol
100:
186-193,
2006.
|
| 148. |
Paydarfar D,
Buerkel DM.
Dysrhythmias of the respiratory oscillator.
Chaos
5:
18-29,
1995.
|
| 149. |
Peng CK,
Buldyrev SV,
Goldberger AL,
Havlin S,
Sciortino F,
Simons M,
Stanley HE.
Long‐range correlations in nucleotide sequences.
Nature
356:
168-170,
1992.
|
| 150. |
Peng CK,
Havlin S,
Stanley HE,
Goldberger AL.
Quantification of scaling exponents and crossover phenomena in nonstationary heartbeat time series.
Chaos
5:
82-87,
1995.
|
| 151. |
Peng CK,
Mietus JE,
Liu Y,
Lee C,
Hausdorff JM,
Stanley HE,
Goldberger AL,
Lipsitz LA.
Quantifying fractal dynamics of human respiration: Age and gender effects.
Ann Biomed Eng
30:
683-692,
2002.
|
| 152. |
Politi AZ,
Donovan GM,
Tawhai MH,
Sanderson MJ,
Lauzon AM,
Bates JH,
Sneyd J.
A multiscale, spatially distributed model of asthmatic airway hyper‐responsiveness.
J Theor Biol
266:
614-624.
|
| 153. |
Poon CS,
Barahona M.
Titration of chaos with added noise.
Proc Natl Acad Sci U S A
98:
7107-7112,
2001.
|
| 154. |
Presson RG Jr,
Okada O,
Hanger CC,
Godbey PS,
Graham JA,
Glenny RW,
Capen RL,
Wagner WW Jr.
Stability of alveolar capillary opening pressures.
J Appl Physiol
77:
1630-1637,
1994.
|
| 155. |
Prigogine I,
Stengers I.
Order Out of Chaos: Man's New Dialogue with Nature.
New York, N.Y.:
Bantam Books,
1984.
|
| 156. |
Que CL,
Kenyon CM,
Olivenstein R,
Macklem PT,
Maksym GN.
Homeokinesis and short‐term variability of human airway caliber.
J Appl Physiol
91:
1131-1141,
2001.
|
| 157. |
Redaelli A,
Vesentini S,
Soncini M,
Vena P,
Mantero S,
Montevecchi FM.
Possible role of decorin glycosaminoglycans in fibril to fibril force transfer in relative mature tendons-a computational study from molecular to microstructural level.
J Biomech
36:
1555-1569,
2003.
|
| 158. |
Ribeiro AS,
Kauffman SA.
Noisy attractors and ergodic sets in models of gene regulatory networks.
J Theor Biol
247:
743-755,
2007.
|
| 159. |
Richter DW,
Ballantyne, D.
A Three Phase Theory About the Basic Respiratory Pattern Generator.
Berlin:
Springer‐Verlag,
1983.
|
| 160. |
Ritter MC,
Jesudason R,
Majumdar A,
Stamenovic D,
Buczek‐Thomas JA,
Stone PJ,
Nugent MA,
Suki B.
A zipper network model of the failure mechanics of extracellular matrices.
Proc Natl Acad Sci U S A
106:
1081-1086,
2009.
|
| 161. |
Robertson HT,
Altemeier WA,
Glenny RW.
Physiological implications of the fractal distribution of ventilation and perfusion in the lung.
Ann Biomed Eng
28:
1028-1031,
2000.
|
| 162. |
Robertson HT,
Hlastala MP.
Microsphere maps of regional blood flow and regional ventilation.
J Appl Physiol
102:
1265-1272,
2007.
|
| 163. |
Romero FJ,
Pastor A,
Lopez J,
Romero PV.
A recruitment‐based rheological model for mechanical behavior of soft tissues.
Biorheology
35:
17-35,
1998.
|
| 164. |
Rostig S,
Kantelhardt JW,
Penzel T,
Cassel W,
Peter JH,
Vogelmeier C,
Becker HF,
Jerrentrup A.
Nonrandom variability of respiration during sleep in healthy humans.
Sleep
28:
411-417,
2005.
|
| 165. |
Sachis PN,
Armstrong DL,
Becker LE,
Bryan AC.
Myelination of the human vagus nerve from 24 weeks postconceptional age to adolescence.
J Neuropathol Exp Neurol
41:
466-472,
1982.
|
| 166. |
Sapoval B,
Filoche M.
Role of diffusion screening in pulmonary diseases.
Adv Exp Med Biol
605:
173-178,
2008.
|
| 167. |
Sapoval B,
Filoche M,
Weibel ER.
Smaller is better—but not too small: A physical scale for the design of the mammalian pulmonary acinus.
Proc Natl Acad Sci U S A
99:
10411-10416,
2002.
|
| 168. |
Sasaki N,
Shukunami N,
Matsushima N,
Izumi Y.
Time‐resolved X‐ray diffraction from tendon collagen during creep using synchrotron radiation.
J Biomech
32:
285-292,
1999.
|
| 169. |
Sato A,
Hirai T,
Imura A,
Kita N,
Iwano A,
Muro S,
Nabeshima Y,
Suki B,
Mishima M.
Morphological mechanism of the development of pulmonary emphysema in klotho mice.
Proc Natl Acad Sci U S A
104:
2361-2365,
2007.
|
| 170. |
Savage VM,
Deeds EJ,
Fontana W.
Sizing up allometric scaling theory.
PLoS Comput Biol
4:
e1000171,
2008.
|
| 171. |
Schmidt‐Nielsen K.
Scaling, why is animal size so important?
Cambridge, New York:
Cambridge University Press,
1984.
|
| 172. |
Schmidt A,
Zidowitz S,
Kriete A,
Denhard T,
Krass S,
Peitgen HO.
A digital reference model of the human bronchial tree.
Comput Med Imaging Graph
28:
203-211,
2004.
|
| 173. |
Seely AJ,
Macklem PT.
Complex systems and the technology of variability analysis.
Crit Care
8:
R367-384,
2004.
|
| 174. |
Shibata H,
Horie O,
Sugita M.
[Studies on the deviation of short‐term FRC measurements with He closed‐circuit method and on diurnal variation of FRC].
Rinsho Byori
38:
463-467,
1990.
|
| 175. |
Shin'oka T,
Matsumura G,
Hibino N,
Naito Y,
Watanabe M,
Konuma T,
Sakamoto T,
Nagatsu M,
Kurosawa H.
Midterm clinical result of tissue‐engineered vascular autografts seeded with autologous bone marrow cells.
J Thorac Cardiovasc Surg
129:
1330-1338,
2005.
|
| 176. |
Small M,
Judd K,
Lowe M,
Stick S.
Is breathing in infants chaotic? Dimension estimates for respiratory patterns during quiet sleep.
J Appl Physiol
86:
359-376,
1999.
|
| 177. |
Stanley HE,
Amaral LA,
Buldyrev SV,
Gopikrishnan P,
Plerou V,
Salinger MA.
Self‐organized complexity in economics and finance.
Proc Natl Acad Sci U S A
99:
(Suppl 1):
2561-2565,
2002.
|
| 178. |
Stanley HE,
Amaral LA,
Goldberger AL,
Havlin S,
Ivanov P,
Peng CK.
Statistical physics and physiology: Monofractal and multifractal approaches.
Physica A
270:
309-324,
1999.
|
| 179. |
Stanley HE,
Buldyrev SV,
Goldberger AL,
Hausdorff JM,
Havlin S,
Mietus J,
Peng CK,
Sciortino F,
Simons M.
Fractal landscapes in biological systems: Long‐range correlations in DNA and interbeat heart intervals.
Physica A
191:
1-12,
1992.
|
| 180. |
Stauffer D,
Aharony A.
Introduction to percolation theory.
London; Washington, DC:
Taylor & Francis,
1992.
|
| 181. |
Suki B.
Fluctuations and power laws in pulmonary physiology.
Am J Respir Crit Care Med
166:
133-137,
2002.
|
| 182. |
Suki B,
Alencar AM,
Sujeer MK,
Lutchen KR,
Collins JJ,
Andrade JS Jr,
Ingenito EP,
Zapperi S,
Stanley HE.
Life‐support system benefits from noise.
Nature
393:
127-128,
1998.
|
| 183. |
Suki B,
Alencar AM,
Tolnai J,
Asztalos T,
Petak F,
Sujeer MK,
Patel K,
Patel J,
Stanley HE,
Hantos Z.
Size distribution of recruited alveolar volumes in airway reopening.
J Appl Physiol
89:
2030-2040,
2000.
|
| 184. |
Suki B,
Barabasi AL,
Hantos Z,
Petak F,
Stanley HE.
Avalanches and power‐law behaviour in lung inflation.
Nature
368:
615-618,
1994.
|
| 185. |
Suki B,
Barabasi AL, and
Lutchen KR.
Lung tissue viscoelasticity: A mathematical framework and its molecular basis.
J Appl Physiol
76:
2749-2759,
1994.
|
| 186. |
Suki B,
Frey U.
Temporal dynamics of recurrent airway symptoms and cellular random walk.
J Appl Physiol
95:
2122-2127,
2003.
|
| 187. |
Suki B,
Lutchen KR,
Ingenito EP.
On the progressive nature of emphysema: Roles of proteases, inflammation, and mechanical forces.
Am J Respir Crit Care Med
168:
516-521,
2003.
|
| 188. |
Suki B,
Majumdar A,
Nugent MA,
Bates JH.
In silico modeling of interstitial lung mechanics: Implications for disease development and repair.
Drug Discov Today Dis Models
4:
139-145,
2007.
|
| 189. |
Szeto HH,
Cheng PY,
Decena JA,
Cheng Y,
Wu DL,
Dwyer G.
Fractal properties in fetal breathing dynamics.
Am J Physiol
263:
R141-147,
1992.
|
| 190. |
Thamrin C,
Stern G,
Strippoli MP,
Kuehni CE,
Suki B,
Taylor DR, and
Frey U.
Fluctuation analysis of lung function as a predictor of long‐term response to beta2‐agonists.
Eur Respir J
33:
486-‐493,
2009.
|
| 191. |
Thamrin C,
Taylor DR,
Jones AD,
Suki B,
Frey U.
Variability of lung function predicts loss of asthma control following withdrawal of inhaled corticosteroid treatment.
Thorax
65(5):
403-‐408,
2010.
|
| 192. |
Troyanov S,
Ghezzo H,
Cartier A,
Malo JL.
Comparison of circadian variations using FEV1 and peak expiratory flow rates among normal and asthmatic subjects.
Thorax
49:
775-780,
1994.
|
| 193. |
Trubel H,
Banikol WK.
Variability analysis of oscillatory airway resistance in children.
Eur J Appl Physiol
94:
364-370,
2005.
|
| 194. |
Tsuda A,
Rogers RA,
Hydon PE,
Butler JP.
Chaotic mixing deep in the lung.
Proc Natl Acad Sci U S A
99:
10173-10178,
2002.
|
| 195. |
Venegas JG,
Winkler T,
Musch G,
Vidal Melo MF,
Layfield D,
Tgavalekos N,
Fischman AJ,
Callahan RJ,
Bellani G,
Harris RS.
Self‐organized patchiness in asthma as a prelude to catastrophic shifts.
Nature
434:
777-782,
2005.
|
| 196. |
Vlahakis NE,
Hubmayr RD.
Cellular stress failure in ventilator‐injured lungs.
Am J Respir Crit Care Med
171:
1328-1342,
2005.
|
| 197. |
Wagner WW Jr,
Todoran TM,
Tanabe N,
Wagner TM,
Tanner JA,
Glenny RW,
Presson RG Jr.
Pulmonary capillary perfusion: Intra‐alveolar fractal patterns and interalveolar independence.
J Appl Physiol
86:
825-831,
1999.
|
| 198. |
Wang Y,
Bai C,
Li K,
Adler KB,
Wang X.
Role of airway epithelial cells in development of asthma and allergic rhinitis.
Respir Med
102:
949-955,
2008.
|
| 199. |
Weibel ER.
Fractal geometry: A design principle for living organisms.
Am J Physiol
261:
L361-369,
1991.
|
| 200. |
Weibel ER.
Morphometry of the human lung.
New York:
Academic Press,
1963.
|
| 201. |
Weibel ER.
What makes a good lung?
Swiss Med Wkly
139:
375-386,
2009.
|
| 202. |
Weiss DJ.
Stem cells and cell therapies for cystic fibrosis and other lung diseases.
Pulm Pharmacol Ther
21:
588-594,
2008.
|
| 203. |
West BJ.
Physiology in fractal dimensions: Error tolerance.
Ann Biomed Eng
18:
135-149,
1990.
|
| 204. |
West GB,
Brown JH,
Enquist BJ.
A general model for the origin of allometric scaling laws in biology.
Science
276:
122-126,
1997.
|
| 205. |
West GB,
Woodruff WH,
Brown JH.
Allometric scaling of metabolic rate from molecules and mitochondria to cells and mammals.
Proc Natl Acad Sci U S A
99:
(Suppl 1):
2473-2478,
2002.
|
| 206. |
White EP,
Enquist BJ,
Green JL.
On estimating the exponent of power‐law frequency distributions.
Ecology
89:
905-912,
2008.
|
| 207. |
Wirtz HR,
Dobbs LG.
Calcium mobilization and exocytosis after one mechanical stretch of lung epithelial cells.
Science
250:
1266-1269,
1990.
|
| 208. |
Witten ML,
Tinajero JP,
Sobonya RE,
Lantz RC,
Quan SF,
Lemen RJ.
Human alveolar fractal dimension in normal and chronic obstructive pulmonary disease subjects.
Res Commun Mol Pathol Pharmacol
98:
221-230,
1997.
|
| 209. |
Wysocki M,
Fiamma MN,
Straus C,
Poon CS,
Similowski T.
Chaotic dynamics of resting ventilatory flow in humans assessed through noise titration.
Respir Physiol Neurobiol
153:
54-65,
2006.
|
| 210. |
Yernault JC,
de Jonghe M,
de Coster A,
Englert M.
Pulmonary mechanics in diffuse fibrosing alveolitis.
Bull Physiopathol Respir (Nancy)
11:
231-244,
1975.
|
| 211. |
Yoo Y,
Choung JT,
Yu J,
Kim do K,
Koh YY.
Acute effects of Asian dust events on respiratory symptoms and peak expiratory flow in children with mild asthma.
J Korean Med Sci
23:
66-71,
2008.
|
| 212. |
Yoo Y,
Kim DK,
Yu J,
Choi SH,
Kim CK,
Koh YY.
Relationships of methacholine and AMP responsiveness with peak expiratory flow variability in children with asthma.
Clin Exp Allergy
37:
1158-1164,
2007.
|
| 213. |
Zamir M.
Fractal dimensions and multifractility in vascular branching.
J Theor Biol
212:
183-190,
2001.
|
| 214. |
Zapperi S,
Ray P,
Stanley HE,
Vespignani A.
Avalanches in breakdown and fracture processes.
Phys Rev E Stat Phys Plasmas Fluids Relat Interdiscip Topics
59:
5049-5057,
1999.
|