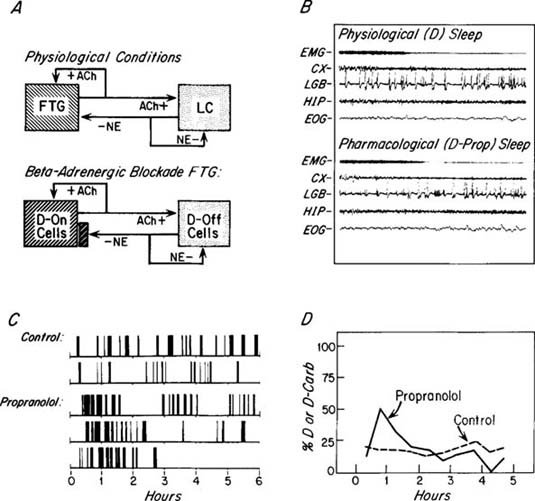
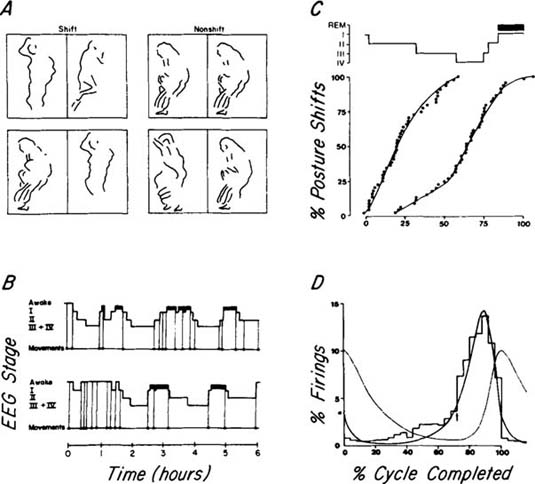
| References |
| 1. |
Aaronson, S. T.,
M. P. Biber,
S. Rashed, and
J. A. Hobson.
Use of time lapse video tape recording in sleep research: method of study and sleep latency determination (Abstract).
Sleep Res.
9:
118,
1980.
|
| 2. |
Aghajanian, G. K.,
J. M. Cederbaum, and
R. Y. Wang.
Evidence for norepinephrine‐mediated collateral inhibition of the locus coeruleus neurons.
Brain Res.
136:
570-577,
1977.
|
| 3. |
Aghajanian, G. K., and
H. J. Haigler.
L‐Tryptophan as a selective histochemical marker for serotonergic neurons in single‐cell recording studies.
Brain Res.
81:
364-372,
1974.
|
| 4. |
Aghajanian, G. K., and
R. Y. Wang.
Habenular and other midbrain raphe afferents demonstrated by a modified retrograde tracing technique.
Brain Res.
122:
229-242,
1977.
|
| 5. |
Aghajanian, G. K., and
R. Y. Wang.
Physiology and pharmacology of central serotonergic neurons. In:
Psychopharmacology: A Generation of Progress,
edited by M. A. Lipton,
A. DiMascio, and
K. F. Killam.
New York:
Raven,
1978,
p. 171-184.
|
| 6. |
Aghajanian, G. K.,
R. Y. Wang, and
J. Baraban.
Serotonergic and nonserotonergic neurons of the dorsal raphe: reciprocal changes in firing induced by peripheral nerve stimulation.
Brain Res.
153:
169-175,
1978.
|
| 7. |
Agid, Y.,
F. Javoy,
J. Glowinski,
D. Bouvet, and
C. Sotelo.
Injection of 6‐hydroxydopamine into the substantia nigra of the rat. II. Diffusion and specificity.
Brain Res.
58:
291-301,
1973.
|
| 8. |
Ahlsén, G., and
S. Lindström.
Excitation of perigeniculate neurones via axon collaterals of principal cells.
Brain Res.
236:
477-481,
1982.
|
| 9. |
Ahlsén, G.,
S. Lindström, and
F. S. Loo.
Functional distinction of perigeniculate and thalamic reticular neurons of the cat.
Exp. Brain Res.
46:
118-126,
1982.
|
| 10. |
Ahlsén, G.,
S. Lindström, and
E. Sybirska.
Subcortical axon collaterals of principal cells in the lateral geniculate body of the cat.
Brain Res.
156:
106-109,
1978.
|
| 11. |
Akindle, M. O.,
J. I. Evans, and
I. Oswald.
Monoamine oxidase inhibitors, sleep and mood.
Electroencephalogr. Clin. Neurophysiol.
29:
47-56,
1970.
|
| 12. |
Albe‐Fessard, D.,
A. Levante, and
Y. Lamour.
Origin of spinothalamic and spinoreticular pathways in cats and monkeys. In:
Advances in Neurology. International Symposium on Pain,
edited by J. J. Bonica.
New York:
Raven,
1974,
p. 157-166.
|
| 13. |
Alger, B. E., and
R. A. Nicoll.
Epileptiform burst after‐hyperpolarization: calcium‐dependent potassium potential in hippocampal CA1 pyramidal cells.
Science
210:
1122-1124,
1980.
|
| 14. |
Allison, T.
Cortical and subcortical evoked responses to central stimuli during wakefulness and sleep.
Electroencephalogr. Clin. Neurophysiol.
18:
131-139,
1965.
|
| 15. |
Allison, T., and
G. D. Goff.
Potentials evoked in somatosensory cortex to thalamo‐cortical radiation stimulation during waking, sleep and arousal from sleep.
Arch. Ital. Biol.
106:
41-60,
1968.
|
| 16. |
Altman, J., and
S. A. Bayer.
Development of the diencephalon in the rat. IV. Quantitative study of the time of origin of neurons and the internuclear chronological gradients in the thalamus.
J. Comp. Neurol.
188:
455-472,
1979.
|
| 17. |
Amaral, D. G., and
H. M. Sinnamon.
The locus coeruleus: neurobiology of a central noradrenergic nucleus.
Prog. Neurobiol.
9:
147-196,
1977.
|
| 18. |
Amassian, V. E., and
R. V. De Vito.
Unit activity in reticular formation and nearby structures.
J. Neurophysiol.
17:
575-603,
1954.
|
| 19. |
Amassian, V. E., and
H. J. Waller.
Spatio‐temporal patterns of activity in individual reticular neurons. In:
Reticular Formation of the Brain,
edited by H. H. Jasper,
L. D. Proctor,
R. S. Knighton,
W. C. Noshay, and
R. T. Costello.
Boston, MA:
Little, Brown,
1958,
p. 69-108.
|
| 20. |
Amatruda, T. T., III,
D. A. Black,
T. M. McKenna,
R. W. McCarley, and
J. A. Hobson.
Sleep cycle control and cholinergic mechanisms: differential effects of carbachol at pontine brain stem sites.
Brain Res.
98:
501-515,
1975.
|
| 21. |
Andén, N. E.,
A. Carlsson,
A. Dahlström,
K. Fuxe,
N. A. Hillarp, and
K. Larsson.
Demonstration and mapping out of nigro‐neostriatal dopamine neurons.
Life Sci.
3:
523-531,
1964.
|
| 22. |
Andén, N. E.,
A. Dahlstrom,
K. Fuxe,
K. Larsson,
L. Olson, and
V. Ungerstedt.
Ascending monoamine neurons to the telencephalon and diencephalon.
Acta Physiol. Scand.
67:
313-326,
1966.
|
| 23. |
Andén, N.,
K. Fuxe,
B. Hamberger, and
T. Hökfelt.
A quantitative study of the nigro‐neostriatal dopamine neuron system in rat.
Acta Physiol. Scand.
67:
306-312,
1966.
|
| 24. |
Andén, N. E.,
M. G. M. Jukes,
A. Lundberg, and
L. Vyklicky.
The effect of DOPA on the spinal cord.
Acta Physiol. Scand.
68:
322-336,
1966.
|
| 25. |
Andersen, P., and
S. A. Andersson.
Physiological Basis of the Alpha Rhythm.
New York:
Appleton‐Century‐Crofts,
1968.
|
| 26. |
Andersen, P.,
S. A. Andersson, and
T. Lomo.
Some factors involved in the thalamic control of spontaneous barbiturate spindles.
J. Physiol. London
192:
257-281,
1967.
|
| 27. |
Andersen, P.,
R. Dingledine,
L. Gjerstad,
I. A. Langmoen, and
A. Laursen‐Mosfeldt.
Two different responses of hippocampal pyramidal cells to application of gamma‐aminobutyric acid.
J. Physiol. London
305:
279-296,
1980.
|
| 28. |
Andersen, P., and
J. C. Eccles.
Inhibitory phasing of neuronal discharge.
Nature London
196:
645-647,
1962.
|
| 29. |
Andersen, P.,
J. C. Eccles, and
T. A. Sears.
The ventrobasal complex of the thalamus: types of cells, their responses and their functional organization.
J. Physiol. London
174:
370-399,
1964.
|
| 30. |
Andersen, P., and
T. A. Sears.
The role of inhibition in the phasing of spontaneous thalamocortical discharge.
J. Physiol. London
173:
459-480,
1964.
|
| 31. |
Anderson, M., and
M. V. Salmon.
Symptomatic cataplexy.
J. Neurol. Neurosurg. Psychiatry
40:
186-191,
1977.
|
| 32. |
Anderson, M. E., and
M. Yoshida.
Axonal branching patterns and location of nigrothalamic and nigrocollicular neurons in the cat.
J. Neurophysiol.
43:
883-895,
1980.
|
| 33. |
Andersson, S. A., and
J. R. Manson.
Rhythmic activity in the thalamus of the unanesthetized decorticate cat.
Electroencephalogr. Clin. Neurophysiol.
31:
21-34,
1971.
|
| 34. |
Angaut, P., and
D. Bowsher.
Ascending projections of the medial cerebellar (fastigial) nucleus: an experimental study in the cat.
Brain Res.
24:
49-68,
1970.
|
| 35. |
Angeleri, F.,
G. F. Marchesi, and
A. Quattrini.
Effects of chronic thalamic lesions on the electrical activity of the neocortex and on sleep.
Arch. Ital. Biol.
107:
633-667,
1969.
|
| 36. |
Apostol, G., and
O. D. Creutzfeldt.
Crosscorrelation between the activity of septal units and hippocampal EEG during arousal.
Brain Res.
67:
65-75,
1974.
|
| 37. |
Araki, T., and
K. Endo.
Short latency EPSPs of pyramidal tract cells evoked by stimulation of the centrum medianum‐parafascicular complex and the nucleus ventralis anterior of the thalamus.
Brain Res.
113:
405-410,
1976.
|
| 38. |
Arduini, A.,
G. Berlucchi, and
P. Strata.
Pyramidal activity during sleep and wakefulness.
Arch. Ital. Biol.
101:
530-544,
1963.
|
| 39. |
Asanuma, H.,
J. Fernandez,
M. E. Scheibel, and
A. B. Scheibel.
Characteristics of projections from the nucleus ventralis lateralis to the motor cortex in the cats: an anatomical and physiological study.
Exp. Brain Res.
20:
315-330,
1974.
|
| 40. |
Aschoff, J.
Circadian Clocks.
Amsterdam:
North‐Holland,
1965.
|
| 41. |
Aschoff, J.
Circadian rhythms in man.
Science
148:
1427-1432,
1965.
|
| 42. |
Aserinsky, E., and
N. Kleitman.
Regularly occurring periods of eye motility and concurrent phenomena during sleep.
Science
118:
273-274,
1953.
|
| 43. |
Aserinsky, E., and
N. Kleitman.
Two types of ocular motility occurring in sleep.
J. Appl. Physiol.
8:
1-10,
1955.
|
| 44. |
Ashby, W. R.
Design for a Brain: The Origin of Adaptive Behaviour
(2nd ed.).
New York:
Wiley,
1960.
|
| 45. |
Ashby, W. R.
Introduction to Cybernetics.
New York:
Science Eds.,
1963.
|
| 46. |
Aston‐Jones, G., and
F. E. Bloom.
Activity of norepinephrine‐containing locus coeruleus neurons in behaving rats anticipates fluctuations in the sleep‐waking cycle.
J. Neurosci.
1:
876-886,
1981.
|
| 47. |
Aston‐Jones, G., and
F. E. Bloom.
Norepinephrine‐containing locus coeruleus neurons in behaving rats exhibit pronounced responses to non‐noxious environmental stimuli.
J. Neurosci.
1:
887-900,
1981.
|
| 48. |
Astruc, J.
Corticofugal fiber degeneration following lesions of area 8 (frontal eye field) in Macaca mulatta (Abstract).
Anat. Rec.
148:
256,
1964.
|
| 49. |
Axelrod, J.
The pineal gland: a neurochemical transducer.
Science
184:
1341-1348,
1974.
|
| 50. |
Baghdoyan, H. A.,
A. P. Monaco,
M. L. Rodrigo‐Angulo,
F. Assens,
R. W. McCarley, and
J. A. Hobson.
Microinjection of neostigmine into the pontine reticular formation of cats enhances desynchronized sleep signs.
J. Pharmacol. Exp. Ther.
231:
173-180,
1984.
|
| 51. |
Baghdoyan, H. A.,
M. L. Rodrigo‐Angulo,
R. W. McCarley, and
J. A. Hobson.
Site‐specific enhancement and suppression of desynchronized sleep signs following cholinergic stimulation of three brainstem regions.
Brain Res.
306:
39-52,
1984.
|
| 52. |
Baker, T. L., and
D. J. McGinty.
Sleep apnea in hypoxic and normal kittens.
Dev. Psychobiol.
12:
577-594,
1979.
|
| 53. |
Baraitser, M., and
J. K. Parkes.
Genetic study of narcoleptic syndrome.
J. Med. Genet.
15:
254-259,
1978.
|
| 54. |
Barchas, J. D.,
W. C. Dement,
D. Faull,
A. S. Foutz, and
R. B. Holzman.
The concentration of amine metabolites in cerebrospinal fluid from normal and narcoleptic dogs.
J. Physiol. London
296:
94-95,
1979.
|
| 55. |
Barrionuevo, G.,
O. Benoit, and
P. Tempier.
Evidence for two types of firing pattern during the sleep‐waking cycle in the reticular thalamic nucleus of the cat.
Exp. Neurol.
72:
486-501,
1981.
|
| 56. |
Bartlett, J. R., and
R. W. Doty, Sr.
Influence of mesencephalic stimulation on unit activity in striate cortex of squirrel monkeys.
J. Neurophysiol.
37:
642-652,
1974.
|
| 57. |
Bartlett, J. R.,
R. W. Doty,
J. Pecci‐Saavedra, and
P. D. Wilson.
Mesencephalic control of lateral geniculate nucleus in primates. III. Modifications with state of alertness.
Exp. Brain Res.
18:
214-224,
1973.
|
| 58. |
Bartolini, A., and
G. Pepeu.
Investigations into the acetylcholine output from the cerebral cortex of the cat in the presence of hyoscine.
Br. J. Pharmacol. Chemother.
31:
66-74,
1967.
|
| 59. |
Batsel, H. L.
Spontaneous desynchronization in the chronic cat “cerveau isolé.”
Arch. Ital. Biol.
102:
547-566,
1964.
|
| 60. |
Baughman, R. W., and
C. D. Gilbert.
Aspartate and glutamate as possible neurotransmitters in the visual cortex.
J. Neurosci.
1:
427-439,
1981.
|
| 61. |
Baxter, B. L.
Induction of both emotional behavior and a novel form of REM sleep by chemical stimulation applied to cat mesencephalon.
Exp. Neurol.
23:
220-230,
1969.
|
| 62. |
Belardetti, F.,
R. Borgia, and
M. Mancia.
Prosencephalic mechanisms of ECoG desynchronization in cerveau isolé cats.
Electroencephahgr. Clin. Neurophysiol.
42:
213-225,
1977.
|
| 63. |
Belin, M. F.,
M. Aguera,
M. Tappaz,
A. McRae‐Dequeurce,
P. Bobillier, and
J. F. Pujol.
GABA‐accumulating neurons in the nucleus raphe dorsalis and periaqueductal gray in the rat: a biochemical and radioautographic study.
Brain Res.
170:
279-297,
1979.
|
| 64. |
Bell, C.,
G. Sierra,
N. Buendia, and
J. P. Segundo.
Sensory properties of neurons in the mesencephalic reticular formation.
J. Neurophysiol.
27:
961-987,
1964.
|
| 65. |
Ben‐Ari, Y.,
R. Dingledine,
I. Kanazawa, and
J. S. Kelly.
Inhibitory effects of acetylcholine on neurons in the feline nucleus reticularis thalami.
J. Physiol. London
261:
647-671,
1976.
|
| 66. |
Ben‐Ari, Y.,
K. Krnjević,
W. Reinhardt, and
N. Ropert.
Intracellular observations on the disinhibitory action of acetylcholine in the hippocampus.
Neuroscience
6:
2475-2484,
1981.
|
| 67. |
Bénita, M., and
H. Condé
Intranuclear organization of the centre median nucleus of thalamus.
J. Physiol. Paris
64:
561-582,
1972.
|
| 68. |
Bentivoglio, M.,
G. Macchi, and
A. Albanese.
The cortical projections of the thalamic intralaminar nuclei, as studied in cat and rat with the multiple fluorescent retrograde tracing technique.
Neurosci. Lett.
26:
5-10,
1981.
|
| 69. |
Bentivoglio, M.,
G. Macchi,
P. Rossini, and
E. Tempesta.
Brain stem neurons projecting to neocortex: an HRP study in the cat.
Exp. Brain Res.
31:
489-498,
1978.
|
| 70. |
Berger, H.
Über das Elektrencephalogram des Menschen. Zweite Mitteilung.
J. Psychol. Neurol.
40:
160-179,
1930.
|
| 71. |
Berger, R. J.
Oculomotor control: a possible function of REM sleep.
Psychol. Rev.
76:
144-164,
1969.
|
| 72. |
Berk, M. L., and
J. A. Finkelstein.
An autoradiographic determination of the efferent projections of the suprachiasmatic nucleus of the hypothalamus.
Brain Res.
226:
1-13,
1981.
|
| 73. |
Berkley, K.
Spatial relationship between the termination of somatic sensory and motor pathways in the rostral brainstem of cats and monkeys. I. Ascending somatic sensory inputs to lateral diencephalon.
J. Comp. Neurol.
193:
283-317,
1980.
|
| 74. |
Berman, A. L.
The Brain Stem of the Cat.
Madison:
Univ. of Wisconsin Press,
1968.
|
| 75. |
Berman, A. L., and
E. G. Jones.
The Thalamus and Basal Telencephalon of the Cat.
Madison:
Univ. of Wisconsin Press,
1982.
|
| 76. |
Berman, N.
Connections of the pretectum in the cat.
J. Comp. Neurol.
174:
227-254,
1977.
|
| 77. |
Berrevoets, C. E., and
H. G. J. M. Kuypers.
Pericruciate cortical neurons projecting to brain stem reticular formation, dorsal column nuclei and spinal cord in the cat.
Neurosci. Lett.
1:
257-262,
1975.
|
| 78. |
Berridge, M. J.
Modulation of nervous activity by cyclic nucleotides and calcium. In:
The Neurosciences: Fourth Study Program,
edited by F. O. Schmitt and
F. G. Worden.
Cambridge, MA:
MIT Press,
1979,
p. 873-889.
|
| 79. |
Besset, A.,
A. Bonardet,
M. Billiard,
B. Descomps,
A. C. De Paulet, and
P. Passouant.
Circadian patterns of growth hormone and Cortisol secretions in narcoleptic patients.
Chronobiologia
6:
19-31,
1979.
|
| 80. |
Besson, J. M.,
G. Guilbaud,
M. Abdelmoumène, and
A. Chaouch.
Physiologie de la nociception.
J. Physiol. Paris
78:
7-107,
1982.
|
| 81. |
Bianchi, C.,
G. Spidelieri,
P. Guandalini,
S. Tangenelli, and
L. Beani.
Inhibition of acetylcholine outflow from the guinea‐pig cerebral cortex following locus coeruleus stimulation.
Neurosci. Lett.
14:
97-100,
1979.
|
| 82. |
Bishop, P. O.
Properties of afferent synapses and sensory neurons in the lateral geniculate neurons.
Int. Rev. Neurobiol.
6:
191-255,
1964.
|
| 83. |
Bishop, P. O.,
W. Burke, and
R. Davis.
Single‐unit recording from antidromically activated optic radiation neurones.
J. Physiol. London
162:
432-450,
1962.
|
| 84. |
Bizzi, E., and
D. C. Brooks.
Functional connections between pontine reticular formation and lateral geniculate nucleus during deep sleep.
Arch. Ital. Biol.
101:
666-680,
1963.
|
| 85. |
Bland, B. H.,
P. Andersen,
T. Ganes, and
O. Sveen.
Automated analysis of rhythmicity of physiologically identified hippocampal formation neurons.
Exp. Brain Res.
38:
205-219,
1980.
|
| 86. |
Bloch, V.
Cerebral activation and memory fixation.
Arch. Ital. Biol.
111:
577-590,
1973.
|
| 87. |
Bloch, V.,
E. Hennevin, and
P. Leconte.
Relationship between paradoxical sleep and memory processes. In:
Brain Mechanisms in Memory and Learning: From the Single Neuron to Man,
edited by M. A. B. Frazier.
New York:
Raven,
1979,
p. 329-343.
|
| 88. |
Block, A. J.,
P. G. Boysen,
J. W. Wynne, and
L. A. Hunt.
Sleep apnea, hypopnea and oxygen desaturation in normal subjects. A strong male predominance.
N. Engl. J. Med.
300:
513-517,
1979.
|
| 89. |
Bloom, F. E.
The role of cyclic nucleotides in central synaptic function.
Rev. Physiol. Biochem. Pharmacol.
74:
1-103,
1975.
|
| 90. |
Bloom, F. E.
Central noradrenergic systems: physiology and pharmacology. In:
Psychopharmacology: A Generation of Progress,
edited by M. A. Lipton,
A. DiMascio, and
K. F. Killam.
New York:
Raven,
1978,
p. 131-141.
|
| 91. |
Bloom, F. E.
Chemical integrative processes in the central nervous system. In:
The Neurosciences: Fourth Study Program,
edited by F. O. Schmitt and
F. G. Worden.
Cambridge, MA:
MIT Press,
1979,
p. 51-58.
|
| 92. |
Bloom, F. E.
Chemical coding: modulation and level setting. Chairman's overview of Part IV. In:
The Reticular Formation Revisited,
edited by J. A. Hobson and
M. A. B. Brazier.
New York:
Raven,
1980,
p. 277-284.
|
| 93. |
Bloom, F. E.
How neurotransmitters may differentiate a cell's role in sensorimotor integration and behavioral state control.
Neurosci. Res. Program Bull.
18:
19-21,
1980.
|
| 94. |
Bloom, F. E.,
B. J. Hoffer, and
G. R. Siggins.
Studies on norepinephrine‐containing afferents to Purkinje cells of rat cerebellum. I. Localization to the fibers and their synapses.
Brain Res.
25:
501-552,
1971.
|
| 95. |
Bloom, F. E.,
B. J. Hoffer, and
G. R. Siggins.
Norepinephrine mediated cerebellar synapses: a model system for neuropsychopharmacology.
Biol. Psychiatry
4:
157-177,
1972.
|
| 96. |
Bobillier, P.,
S. Sequin,
F. Petitjean,
D. Salvert,
M. Touret, and
M. Jouvet.
The raphe nuclei of cat brain stem: a topographical atlas of their efferent projections as revealed by autoradiography.
Brain Res.
113:
449-486,
1976.
|
| 97. |
Boivie, J.
The termination of the spinothalamic tract in the cat. An experimental study with silver impregnation methods.
Exp. Brain Res.
12:
331-353,
1971.
|
| 98. |
Bolam, J. P.,
P. Somogyi,
S. Totterdell, and
A. D. Smith.
A second type of striatonigral neuron: a comparison between retrogradely labelled and Golgi‐stained neurons at the light and electron microscopic level.
Neuroscience
6:
2141-2157,
1981.
|
| 99. |
Bouyer, J. J.,
M. F. Montaron, and
A. Rougeul.
Fast fronto‐parietal rhythms during combined focused attentive behaviour and immobility in cat: cortical and thalamic localizations.
Electroencephahgr. Clin. Neurophysiol.
51:
244-252,
1981.
|
| 100. |
Bowsher, D.
Some afferent and efferent connections of the parafascicular‐center median complex. In:
The Thalamas,
edited by D. P. Purpura and
M. D. Yahr.
New York:
Columbia Univ. Press,
1966,
p. 99-108.
|
| 101. |
Bowsher, D., and
J. Westman.
The gigantocellular reticular region and its spinal afferents: a light and electron microscope study in the cat.
J. Anat.
106:
23-36,
1970.
|
| 102. |
Boyle, R., and
O. Pompeiano.
Relation between cell size and response characteristics of vestibulospinal neurons to labyrinth and neck inputs.
J. Neurosci.
1:
1052-1066,
1981.
|
| 103. |
Bremer, F.
Cerveau isolé et physiologie du commeil.
C. R. Soc. Biol.
118:
1235-1241,
1935.
|
| 104. |
Bremer, F.
L'activité cérébrale au cours du sommeil et de la narcose. Contribution à l'étude du mécanisme du sommeil.
Bull. Acad. R. Med. Belg.
4:
68-86,
1937.
|
| 105. |
Bremer, F.
Cerebral hypnogenic centers.
Ann. Neurol.
2:
1-6,
1977.
|
| 106. |
Bremer, F., and
J. Chattonnet.
Acétylcholine et cortex cérébral.
Arch. Int. Physiol.
57:
106-109,
1949.
|
| 107. |
Bremer, F., and
N. Stoupel.
Facilitation et inhibition des potentiels évoqués corticaux dans l'éveil cérébral.
Arch. Int. Pharmacodyn. Ther.
67:
240-275,
1959.
|
| 108. |
Bremer, F., and
C. Terzuolo.
Contribution à l'étude des mécanismes physiologiques du maintien de l'activité vigile du cerveau. Interaction de la formation réticulée et de l'écorce cérébrale dans le processus de l'éveil.
Arch. Int. Pharmacodyn. Ther.
62:
157-178,
1954.
|
| 109. |
Brodal, A.
The Reticular Formation of the Brain Stem: Anatomical Aspects and Functional Correlations.
Edinburgh:
Oliver & Boyd,
1957.
|
| 110. |
Brodal, A., and
G. F. Rossi.
Ascending fibers in brain stem reticular formation of cat.
Arch. Neurol. Psychiatry
74:
68-87,
1955.
|
| 111. |
Bromberg, M. B.,
J. B. Penney, Jr.,
B. S. Stephenson, and
A. B. Young.
Evidence for glutamate as neurotransmitter of corticothalamic and corticorubral pathways.
Brain Res.
215:
369-374,
1981.
|
| 112. |
Brookhart, J. M.
A study of cortico‐spinal activation of motoneurons.
Res. Publ. Assoc. Res. Nerv. Ment. Dis.
30:
157-173,
1950.
|
| 113. |
Brooks, D. C., and
E. Bizzi.
Brain‐stem electrical activity during deep sleep.
Arch. Ital. Biol.
101:
648-665,
1963.
|
| 114. |
Brooks, D. C., and
M. D. Gershon.
Eye movement potentials in the oculomotor and visual systems of the cat: a comparison of reserpine induced waves with those present during wakefulness and rapid eye movement sleep.
Brain Res.
27:
223-239,
1971.
|
| 115. |
Broughton, R. J.
Sleep disorders: disorders of arousal?
Science
159:
1070-1078,
1968.
|
| 116. |
Bunney, B. S., and
G. K. Aghajanian.
The precise localization of nigral afferents in the rat as determined by a retrograde tracing technique.
Brain Res.
117:
423-435,
1976.
|
| 117. |
Burke, R. E.,
R. P. Dum,
J. W. Fleshman,
L. L. Glenn,
A. Lev‐Tov,
M. J. O'Donovan, and
M. J. Pinter.
An HRP study of the relation between cell size and motor unit type in cat ankle extensor motoneurons.
J. Comp. Neurol.
209:
17-28,
1982.
|
| 118. |
Burke, W., and
A. M. Cole.
Extraretinal influences on the lateral geniculate nucleus.
Rev. Physiol. Biochem. Pharmacol.
80:
105-166,
1978.
|
| 119. |
Burke, W., and
A. J. Sefton.
Inhibitory mechanisms in lateral geniculate nucleus of rat.
J. Physiol. London
187:
231-246,
1966.
|
| 120. |
Burns, B. D., and
A. C. Webb.
Mechanisms governing the relation between activity in the cerebral cortex and level of arousal.
Proc. R. Soc. London Ser. B
214:
325-334,
1982.
|
| 121. |
Buser, P., and
F. E. Horvath.
Thalamo‐caudate‐cortical relationships in synchronized activity. II. Further differentiation between spindle systems by cooling and lesions in the mesencephalon.
Brain Res.
39:
43-60,
1972.
|
| 122. |
Butcher, L. L., and
J. Engel.
Behavioral and biochemical effects on L‐dopa after peripheral decarboxylase inhibition.
Brain Res.
15:
233-242,
1969.
|
| 123. |
Büttner‐Ennever, J. A.
Pathways from the pontine reticular formation to structures controlling horizontal and vertical eye movements in the monkey. In:
Control of Gaze by Brainstem Neurons,
edited by R. Baker and
A. Berthoz.
Amsterdam:
Elsevier,
1977,
p. 89-98.
|
| 124. |
Buttner‐Ennever, J. A., and
V. Henn.
An autoradiographic study of the pathways from the pontine reticular formation involved in horizontal eye movements.
Brain Res.
108:
155-164,
1976.
|
| 125. |
Candia, O.,
E. Favale,
A. Guissani, and
G. F. Rossi.
Blood pressure during natural sleep and during sleep induced by electrical stimulation of the brain stem reticular formation.
Arch. Ital. Biol.
100:
216-233,
1962.
|
| 126. |
Card, J. P.,
N. Brecha,
H. J. Karten, and
R. Y. Moore.
Immunocytochemical localization of vasoactive intestinal polypeptide‐containing cells and processes in the suprachiasmatic nucleus of the rat: light and electron microscopic analysis.
J. Neurosci.
1:
1289-1303,
1981.
|
| 127. |
Carman, J. B.,
W. M. Cowan, and
T. P. S. Powell.
Cortical connexions of the thalamic reticular nucleus.
J. Anat.
98:
587-598,
1964.
|
| 128. |
Carter, D. A., and
H. C. Fibiger.
Ascending projections of presumed dopamine‐containing neurons in the ventral tegmentum of the rat as demonstrated by horseradish peroxidase.
Neuroscience
2:
569-576,
1977.
|
| 129. |
Carter, D. A., and
H. C. Fibiger.
The projections of the entopeduncular nucleus and globus pallidus in rat as demonstrated by autoradiography and horseradish peroxidase histochemistry.
J. Comp. Neurol.
177:
113-124,
1978.
|
| 130. |
Castaigne, P.,
A. Buge,
R. Escourolle, and
M. Masson.
Ramollissement pédonculaire médian, tegmento‐thalamique avec ophthalmoplégie et hypersomnie.
Rev. Neurol.
106:
357-367,
1962.
|
| 131. |
Castiglione, A. J.,
M. C. Gallaway, and
J. D. Coulter.
Spinal projections from the midbrain in monkey.
J. Comp. Neurol.
178:
329-346,
1978.
|
| 132. |
Catsman‐Berrevoets, C. E., and
H. G. J. M. Kuypers.
A search for corticospinal collaterals to the thalamus and mesencephalon by means of multiple retrograde fluorescent tracers in cat and rat.
Brain Res.
218:
15-33,
1981.
|
| 133. |
Caviness, V. S., Jr., and
D. O. Frost.
Tangential organization of thalamic projections to the neocortex in the mouse.
J. Comp. Neurol.
194:
335-367,
1980.
|
| 134. |
Cederbaum, J. M., and
G. K. Aghajanian.
Afferent projections to the rat locus coeruleus as determined by a retrograde tracing technique.
J. Comp. Neurol.
178:
1-16,
1978.
|
| 135. |
Celesia, G. G., and
H. H. Jasper.
Acetylcholine released from cerebral cortex in relation to state of activation.
Neurology
16:
1053-1064,
1966.
|
| 136. |
Cesaro, P.,
J. Nguyen‐Legros,
B. Berger,
C. Alvarez, and
D. Albe‐Fessard.
Double labelling of branched neurons in the central nervous system of the rat by retrograde axonal transport of horseradish peroxidase and iron dextran complex.
Neurosci. Lett.
15:
1-7,
1979.
|
| 137. |
Cespuglio, R.,
H. Faradji,
M. E. Gomez, and
M. Jouvet.
Single unit recordings in the nuclei raphe dorsalis and magnus during the sleep‐waking cycle of semi‐chronic prepared cats.
Neurosci. Lett.
24:
133-138,
1981.
|
| 138. |
Cespuglio, R.,
M. E. Gomez,
E. Walker, and
M. Jouvet.
Effets du refroidissement et de la stimulation des noyaux du système du raphé sur les états de vigilance chez le chat.
Electroencephalogr. Clin. Neurophysiol.
47:
289-308,
1979.
|
| 139. |
Cespuglio, R.,
F. Rion,
M. Buda,
H. Faradji,
F. Gonon,
M. Jouvet, and
J. F. Pujol.
Mesure in vivo, par voltamétrie impulsionelle différentielle, du 5‐HIAA dans le striatum du rat.
C. R. Acad. Sci.
290:
901-906,
1980.
|
| 140. |
Chalmers, J. P.,
R. J. Baldessarini, and
R. J. Wurtman.
Effects of L‐Dopa on norepinephrine metabolism in the brain.
Proc. Natl. Acad. Sci. USA
68:
662-666,
1971.
|
| 141. |
Chase, M. H.,
S. H. Chandler, and
Y. Nakamura.
Intracellular determination of membrane potential of trigeminal motoneurons during sleep and wakefulness.
J. Neurophysiol.
44:
349-358,
1980.
|
| 142. |
Cheney, D. L.,
H. F. Le Feure, and
G. Racagni.
Choline acetyltransferase activity and mass fragmentographic measurement of acetylcholine in specific nuclei and tracts of rat brain.
Neuropharmacology
14:
801-809,
1975.
|
| 143. |
Chi, C. C., and
J. P. Flynn.
Neuroanatomic projections related to biting attack elicited from hypothalamus in cats.
Brain Res.
35:
49-66,
1971.
|
| 144. |
Chu, N. S., and
F. E. Bloom.
Norepinephrine‐containing neurons: changes in spontaneous discharge patterns during sleeping and waking.
Science
179:
908-910,
1973.
|
| 145. |
Chu, N. S., and
F. E. Bloom.
Activity patterns of catecholamine‐containing pontine neurons in the dorsolateral tegmentum of unrestrained cats.
J. Neurobiol.
5:
527-544,
1974.
|
| 146. |
Chu, N. S., and
F. E. Bloom.
The catecholamine‐containing neurons in the cat dorsolateral pontine tegmentum: distribution of the cell bodies and some axonal projections.
Brain Res.
66:
1-21,
1974.
|
| 147. |
Churukanov, V.,
B. Pollin, and
D. Albe‐Fessard.
Inhibition d'activités thalamiques par la stimulation du noyau inférieur du raphe chez le chat éveillé chronique.
C. R. Acad. Sci.
283:
1651-1654,
1976.
|
| 148. |
Clamann, H. P., and
E. Hennemann.
Electrical measurement of axon diameter and its use in relating motoneuron size to critical firing level.
J. Neurophysiol.
39:
844-851,
1976.
|
| 149. |
Clark, R. W.,
H. Boudoulas,
S. F. Schaal, and
H. S. Schmidt.
Adrenergic hyperactivity and cardiac abnormality in primary disorders of sleep.
Neurology
30:
113-119,
1980.
|
| 150. |
Coenen, A. M. L., and
A. J. H. Vendrik.
Determination of the transfer ratio of cat's geniculate neurons through quasi‐intracellular recordings and the relation with the level of alertness.
Exp. Brain Res.
14:
227-242,
1972.
|
| 151. |
Cohen, B., and
V. Henn.
Unit activity in the pontine reticular formation associated with eye movement.
Brain Res.
46:
403-410,
1972.
|
| 152. |
Collier, B., and
J. F. Mitchell.
The central release of acetylcholine during consciousness and after brain lesions.
J. Physiol. London
188:
83-99,
1967.
|
| 153. |
Colonnier, M. L.
The structural design of the neocortex. In:
Brain and Conscious Experience,
edited by J. C. Eccles.
New York:
Springer‐Verlag,
1966,
p. 1-23.
|
| 154. |
Colonnier, M.,
M. Steriade, and
P. Landry.
Selective resistance of sensory cells of the mesencephalic trigeminal nucleus to kainic acid‐induced lesions.
Brain Res.
172:
552-556,
1979.
|
| 155. |
Condé, F., and
H. Condé
Observations on the orthograde and retrograde transport of horseradish peroxidase in the cat.
J. Hirnforsch.
20:
35-46,
1979.
|
| 156. |
Coombs, J. S.,
J. C. Eccles, and
P. Fatt.
The electrical properties of the motoneurone membrane.
J. Physiol. London
130:
291-325,
1955.
|
| 157. |
Cordeau, J. P.,
J. De Champlain, and
B. Jacks.
Excitation and prolonged waking produced by catecholamines injected into the ventricular system of cats.
Can. J. Physiol. Pharmacol.
49:
627-631,
1971.
|
| 158. |
Coulter, J. D.,
B. K. Lester, and
H. L. Williams.
Reserpine and sleep.
Psychopharmacologia
19:
134-147,
1971.
|
| 159. |
Coyle, J. T.,
M. E. Molliver, and
M. J. Kuhar.
In situ injection of kainic acid: a new method for selectively lesioning neuronal cell bodies while sparing axons of passage.
J. Comp. Neurol.
180:
301-324,
1978.
|
| 160. |
Cramer, H., and
W. Kuhlo.
Effets des inhibiteurs de la mono‐aminoxydase sur le sommeil et l'électroencephalogramme chez l'homme.
Acta Neurol. Belg.
67:
658-669,
1967.
|
| 161. |
Crawford, J. M., and
D. R. Curtis.
Pharmacological studies on feline Betz cells.
J. Physiol. London
186:
121-138,
1966.
|
| 162. |
Creutzfeldt, O. D.,
S. Watanabe, and
H. D. Lux.
Relations between EEG phenomena and potentials of single cortical cells. I. Evoked responses after thalamic and epicortical stimulation.
Electroencephalogr. Clin. Neurophysiol.
20:
1-18,
1966.
|
| 163. |
Crow, T. J.,
J. F. Deakin,
S. E. File,
A. Longden, and
S. Wendlandt.
The locus coeruleus noradrenergic system—evidence against a role in attention, habituation, anxiety and motor activity.
Brain Res.
155:
249-261,
1978.
|
| 164. |
Crutcher, M. D.,
M. H. Branch,
M. R. De Long, and
A. P. Georgopoulos.
Activity of zona incerta neurons in the behaving primate.
Soc. Neurosci. Abstr.
6:
676,
1980.
|
| 165. |
Cummings, J. P., and
D. L. Felten.
A raphe dendrite bundle in the rabbit medulla.
J. Comp. Neurol.
183:
1-24,
1979.
|
| 166. |
Dagnino, N.,
E. Favale,
C. Loeb, and
M. Manfredi.
Sensory transmission in the geniculostriate system of the cat during natural sleep and arousal.
J. Neurophysiol.
28:
443-456,
1965.
|
| 167. |
Dagnino, N.,
E. Favale,
M. Manfredi,
A. Seitun, and
A. Tartaglione.
Tonic changes in excitability of thalamocortical neurons during the sleep‐waking cycle.
Brain Res.
29:
354-357,
1971.
|
| 168. |
Dahlström, A., and
K. Fuxe.
Evidence for the existence of monoamine neurons in the central nervous system. I. Demonstration of monoamines in the cell bodies of brain stem neurons.
Acta Physiol. Scand. Suppl.
232:
1-55,
1964.
|
| 169. |
Darke, P. G., and
V. Jessen.
Narcolepsy in a dog.
Vet. Rec.
101:
117-118,
1977.
|
| 170. |
Darwin, C.
The Origin of Species by Means of Natural Selection.
London:
Murray,
1878.
|
| 171. |
Dell, P.
Some basic mechanisms of the translation of bodily needs into behaviour. In:
Neurological Basis of Behaviour,
edited by G. E. W. Wolstenholme and
C. M. O'Connor.
London:
Churchill,
1957,
p. 187-203.
|
| 172. |
De Long, M. R.
Activity of pallidal neurons during movement.
J. Neurophysiol.
34:
414-427,
1971.
|
| 173. |
De Long, M. R.
Activity of basal ganglia neurons during movement.
Brain Res.
40:
127-135,
1972.
|
| 174. |
Delorme, F.,
M. Jeannerod, and
M. Jouvet.
Effets remarquables de la réserpine sur l'activité EEG phasique pontogeniculo‐occipitale.
C. R. Soc. Biol.
159:
900-903,
1965.
|
| 175. |
Delphs, J. R., and
M. A. Dichter.
Effects of somastatin on mammalian cortical neurons in culture: physiological actions and unusual dose response characteristics.
J. Neurosci.
3:
1176-1188,
1983.
|
| 176. |
Dement, W.
The occurrence of low voltage, fast, electroencephalogram patterns during behavioral sleep in the cat.
Electroencephalogr. Clin. Neurophysiol.
10:
291-296,
1958.
|
| 177. |
Dement, W. C.,
S. Hendriksen,
B. L. Jacobs, and
M. M. Mitler.
Biogenic amines, phasic events, and behavior. In:
Pharmacology and the Future of Man. Brain, Nerves, and Synapses,
edited by F. E. Bloom and
G. H. Acheson.
New York:
Karger,
1973,
vol. 4,
p. 74-89.
|
| 178. |
Dement, W., and
N. Kleitman.
Cyclic variations in EEG during sleep and their relation to eye movements, body mobility and dreaming.
Electroencephalogr. Clin. Neurophysiol.
9:
673-690,
1957.
|
| 179. |
Dement, W., and
N. Kleitman.
The relation of eye movements during sleep to dream activity: an objective method for the study of dreaming.
J. Exp. Psychol.
53:
89-97,
1957.
|
| 180. |
De Montigny, C., and
J. P. Lund.
Microiontophoretic study of the action of kainic acid and putative transmitters in the rat mesencephalic trigeminal nucleus.
Neuroscience
5:
1621-1628,
1980.
|
| 181. |
Deniau, J. M.,
A. M. Thierry, and
J. Feger.
Electrophysiological identication of mesencephalic ventromedial tegmental (VMT) neurons projecting to the frontal cortex, septum and nucleus accumbens.
Brain Res.
189:
315-326,
1980.
|
| 182. |
Descarries, L.,
A. Beaudet,
K. C. Watkins, and
S. Garcia.
The serotonin neurons in nucleus raphe dorsalis of adult rat (Abstract).
Anat. Rec.
193:
520,
1979.
|
| 183. |
Descarries, L.,
K. C. Watkins, and
Y. Lapierre.
Noradrenergic axon terminals in the cerebral cortex of rat. III. Topometric ultrastructural analysis.
Brain Res.
133:
197-222,
1977.
|
| 184. |
Deschěnes, M.,
M. Paradis,
J. P. Roy, and
M. Steriade.
Electrophysiology of neurons of lateral thalamic nuclei in cat: resting properties and burst discharges.
J. Neurophysiol.
51:
1196-1219,
1984.
|
| 185. |
Deschěnes, M.,
J. P. Roy, and
M. Steriade.
Thalamic bursting mechanism: an inward slow current revealed by membrane hyperpolarization.
Brain Res.
239:
289-293,
1982.
|
| 186. |
Deschěnes, M.,
J. P. Roy, and
M. Steriade.
Nature of 7-14 Hz rhythmic hyperpolarizations in thalamic relay neurons.
Soc. Neurosci. Abstr.
9:
678,
1983.
|
| 187. |
Desmedt, J. E., and
J. Debecker.
Wave form and neural mechanism of the decision P350 elicited without pre‐stimulus CNV or readiness potential in random sequences of near‐threshold auditory clicks and finger stimuli.
Electroencephalogr. Clin. Neurophysiol.
47:
648-670,
1979.
|
| 188. |
Desmedt, J. E., and
D. Robertson.
Differential enhancement of early and late components of the cerebral somatosensory evoked potentials during forced‐pace cognitive tasks in man.
J. Physiol. London
271:
761-782,
1977.
|
| 189. |
Dewan, E. M.
The Programming (P) Hypothesis for REMs.
Washington, DC:
Office of Aerospace Res., USAF,
1969.
(Phys. Sci. Res. Paper 388.)
|
| 190. |
Dierickx, K., and
F. Vandesande.
Immunocytochemical localization of somatostatin‐containing neurons in the rat hypothalamus.
Cell Tissue Res.
201:
349-359,
1979.
|
| 191. |
Dingledine, R., and
J. S. Kelly.
Brain stem stimulation and the acetylcholine‐evoked inhibition of neurones in the feline nucleus reticularis thalami.
J. Physiol. London
271:
135-154,
1977.
|
| 192. |
Dismukes, R. K.
New concepts of molecular communication among neurons.
Behav. Brain Sci.
2:
409-448,
1979.
|
| 193. |
Divac, I.
Magnocellular nuclei of the basal forebrain project to neocortex, brain stem, and olfactory bulb. Review of some functional correlates.
Brain Res.
93:
385-398,
1975.
|
| 194. |
Domer, F. R., and
W. Feldberg.
Some central actions of adrenaline and noradrenaline when administered into the cerebral ventricles. In:
Adrenergic Mechanisms,
edited by G. E. W. Wolstenholme and
M. J. O'Connor.
London:
Churchill,
1960,
p. 386-392.
(Ciba Found. Symp.)
|
| 195. |
Domino, E. F., and
M. Stawiski.
Modification of the cat sleep cycle by hemicholinium‐3, a cholinergic antisynthesis agent.
Res. Commun. Chem. Pathol. Pharmacol.
2:
461-467,
1971.
|
| 196. |
Domino, E. F., and
K. Yamamoto.
Nicotine effect on the sleep cycle of the cat.
Science
150:
637-638,
1965.
|
| 197. |
Domino, E. F.,
K. Yamamoto, and
A. T. Dren.
Role of cholinergic mechanisms in state of wakefulness and sleep. In:
Progress in Brain Research. Anticholinergic Drugs,
edited by P. B. Bradley and
M. Fink.
Amsterdam:
Elsevier,
1968,
vol. 28,
p. 113-133.
|
| 198. |
Donoghue, J. P., and
F. F. Ebner.
The laminar distribution and ultrastructure of fibers projecting from three thalamic nuclei to the somatic sensory‐motor cortex of the opossum.
J. Comp. Neurol.
198:
389-420,
1981.
|
| 199. |
Doty, R. W.
Electrical stimulation of the brain in behavioral context.
Annu. Rev. Psychol.
20:
289-320,
1969.
|
| 200. |
Dubin, M. W., and
B. G. Cleland.
Organization of visual inputs to interneurons of lateral geniculate nucleus of the cat.
J. Neurophysiol.
40:
410-427,
1977.
|
| 201. |
Dumont, S., and
P. Dell.
Facilitation réticulaire des mécanismes visuels corticaux.
Electroencephalogr. Clin. Neurophysiol.
12:
769-796,
1958.
|
| 202. |
Dunleavy, D. L. F.,
V. Brezinova,
I. Oswald,
A. W. MacLean, and
M. Tinkler.
Changes during weeks in effects of tricyclic drugs on the human sleeping brain.
Br. J. Psychiatry.
120:
663-672,
1972.
|
| 203. |
Eccles, J. C.
The Physiology of Synapses.
Berlin:
Springer‐Verlag,
1964.
|
| 204. |
Eccles, J. C.,
R. A. Nicoll,
D. W. F. Schwarz,
H. Táboríková, and
T. J. Willey.
Reticulospinal neurons with and without monosynaptic inputs from cerebellar nuclei.
J. Neurophysiol.
38:
513-530,
1975.
|
| 205. |
Eccles, J. C.,
R. A. Nicoll,
H. Táboríková, and
T. J. Willey.
Medial reticular neurons projecting rostrally.
J. Neurophysiol.
38:
531-538,
1975.
|
| 206. |
Edley, S. M., and
A. M. Graybiel.
The afferent and efferent connections of the feline nucleus tegmenti pedunculopontinus, pars compacta.
J. Comp. Neurol.
217:
187-215,
1983.
|
| 207. |
Edwards, S. B.
Autoradiographic studies of the projections of the midbrain reticular formation: descending projections of nucleus cuneiformis.
J. Comp. Neurol.
161:
341-358,
1975.
|
| 208. |
Edwards, S. B.
The deep cell layers of the superior colliculus: their reticular characteristics and structural organization. In:
The Reticular Formation Revisited,
edited by J. A. Hobson and
M. A. B. Brazier.
New York:
Raven,
1980,
p. 193-209.
|
| 209. |
Edwards, S. B., and
J. S. De Olmos.
Autoradiographic studies of the projections of the midbrain reticular formation: ascending projections of nucleus cuneiformis.
J. Comp. Neurol.
165:
417-432,
1976.
|
| 210. |
Egan, T. M.,
G. Henderson,
R. A. North, and
J. T. Williams.
Noradrenaline‐mediated synaptic inhibition in rat locus coeruleus neurones.
J. Physiol. London
345:
477-488,
1983.
|
| 211. |
Emson, P.C.,
G. Paxinos,
G. Le Gal La Salle,
Y. Ben‐Ari, and
A. Silver.
Choline acetyltransferase and acetylcholinesterase containing projections from the basal forebrain to the amygdaloid complex of the rat.
Brain Res.
165:
271-282,
1979.
|
| 212. |
Endo, K.,
T. Araki, and
K. Ito.
Short latency EPSPs and incrementing PSPs of pyramidal tract cells evoked by stimulation of the nucleus centralis lateralis of the thalamus.
Brain Res.
132:
541-546,
1977.
|
| 213. |
Evarts, E. V.
Effects of sleep and waking on spontaneous and evoked discharge of single units in visual cortex.
Federation Proc. Suppl.
4:
828-837,
1960.
|
| 214. |
Evarts, E. V.
Temporal patterns of discharge of pyramidal tract neurons during sleep and waking in the monkey.
J. Neurophysiol.
27:
152-171,
1964.
|
| 215. |
Evarts, E. V.
Relation of discharge frequency to conduction velocity in pyramidal tract neurons.
J. Neurophysiol.
28:
216-228,
1965.
|
| 216. |
Everett, G. M., and
J. W. Borchering.
L‐Dopa: effect on concentrations of dopamine, norepinephrine and serotonin in brains of mice.
Science
168:
849-850,
1970.
|
| 217. |
Facon, E.,
M. Steriade, and
N. Wertheim.
Hypersomnie prolongée engendrée par des lésions bilatérales du système activateur médial. Le syndrome thrombotique de la bifurcation du tronc basilaire.
Rev. Neurol.
98:
117-133,
1958.
|
| 218. |
Falck, B.,
N. A. Hillarp,
G. Thieme, and
A. Torp.
Fluorescence of catecholamines and related compounds condensed with formaldehyde.
J. Histochem. Cytochem.
10:
348-354,
1962.
|
| 219. |
Fallon, J. H.
Collateralization of monoamine neurons: mesotelencephalic dopamine projections to caudate, septum, and frontal cortex.
J. Neurosci.
1:
1361-1368,
1981.
|
| 220. |
Fallon, J. H., and
R. Y. Moore.
Catecholamine innervation of the basal forebrain. IV. Topography of the dopamine projection to the basal forebrain and neostriatum.
J. Comp. Neurol.
180:
545-580,
1978.
|
| 221. |
Farber, J.,
J. D. Miller,
P. Gatz,
D. C. German, and
H. P. Roffwarg.
Unit activity of CA and non‐CA neurons in midbrain (DA) and hindbrain (NE) in relation to sleep‐wake states (Abstract).
Sleep Res.
10:
27,
1981.
|
| 222. |
Favale, E.,
C. Loeb,
M. Manfredi, and
G. Sacco.
Somatic afferent transmission and cortical responsiveness during natural sleep and arousal in the cat.
Electroencephalogr. Clin. Neurophysiol.
18:
354-368,
1965.
|
| 223. |
Fedina, L.,
A. Lundberg, and
L. Vyklický
The effect of a noradrenaline liberator (4, α‐dimethyl‐meta‐tyramine) on reflex transmission in spinal cats.
Acta Physiol. Scand.
83:
495-504,
1971.
|
| 224. |
Feldberg, W., and
S. L. Sherwood.
Injections of drugs into the lateral ventricle of the cat.
J. Physiol. London
123:
148-167,
1954.
|
| 225. |
Feldman, S. M., and
H. J. Waller.
Dissociation of electrocortical activation and behavioural arousal.
Nature London
196:
1320-1322,
1962.
|
| 226. |
Ferster, D., and
S. Levay.
The axonal arborizations of lateral geniculate neurons in the striate cortex of the cat.
J. Comp. Neurol.
182:
923-944,
1978.
|
| 227. |
Fertziger, A. P., and
D. P. Purpura.
Diphasic‐PSPs during maintained activity of cat lateral geniculate neurons.
Brain Res.
33:
463-467,
1971.
|
| 228. |
Filion, M., and
C. Harnois.
A comparison of projections of entopeduncular neurons to the thalamus, the midbrain and the habenula in the cat.
J. Comp. Neurol.
181:
763-780,
1978.
|
| 229. |
Filion, M.,
Y. Lamarre, and
J. P. Cordeau.
Neuronal discharges of the ventrolateral nucleus of the thalamus during sleep and wakefulness in the cat. II. Evoked activity.
Exp. Brain Res.
12:
499-508,
1971.
|
| 230. |
Fishbein, W., and
B. M. Gutwein.
Paradoxical sleep and memory storage processes.
Behav. Biol.
19:
425-464,
1977.
|
| 231. |
Flicker, C.,
R. W. McCarley, and
J. A. Hobson.
Aminergic neurons: state control and plasticity in three model systems.
Cell. Mol. Neurobiol.
1:
123-166,
1981.
|
| 232. |
Foote, S. L.
Compensatory changes in REM sleep time of cats during ad libitum sleep and following brief REM sleep deprivation.
Brain Res.
54:
261-276,
1973.
|
| 233. |
Foote, S. L.,
G. Aston‐Jones, and
F. E. Bloom.
Impulse activity of locus coeruleus neurons in awake rats and monkeys is a function of sensory stimulation and arousal.
Proc. Natl. Acad. Sci. USA
77:
3033-3037,
1980.
|
| 234. |
Foote, S. L., and
F. E. Bloom.
Activity of norepinephrine‐containing locus coeruleus neurons in the unanesthetized squirrel monkey. In:
Catecholamines: Basic and Clinical Frontiers,
edited by E. Usdin,
I. J. Kopin, and
J. Barchas.
New York:
Pergamon,
1979,
p. 625-627.
|
| 235. |
Foote, S. L.,
F. E. Bloom, and
G. Aston‐Jones.
Nucleus locus ceruleus: new evidence of anatomical and physiological specificity.
Physiol. Rev.
63:
844-914,
1983.
|
| 236. |
Foote, S. L.,
R. Freedman, and
A. P. Olivier.
Effects of putative neurotransmitters on neuronal activity in monkey auditory cortex.
Brain Res.
86:
229-242,
1975.
|
| 237. |
Foster, J. A.
Intracortical origin of recruiting responses in the cat cortex.
Electroencephalogr. Clin. Neurophysiol.
48:
639-653,
1980.
|
| 238. |
Foulkes, D.
The Psychology of Sleep.
New York:
Scribner,
1966.
|
| 239. |
Fourment, A., and
J. C. Hirsch.
Single‐unit discharges in the dorsolateral thalamus of behaving cats: spontaneous activity.
Exp. Neurol.
65:
1-15,
1979.
|
| 240. |
Fram, D. H.,
D. L. Murphy,
F. K. Goodwin,
H. Keith,
H. Brodie,
W. E. Bunney, Jr., and
F. Snyder.
L‐Dopa and sleep in depressed patients.
Psychophysiology
7:
316-317,
1970.
|
| 241. |
Frigyesi, T. L.
Intracellular recordings from neurons in dorsolateral thalamic reticular nucleus during capsular, basal ganglia and midline thalamic stimulation.
Brain Res.
48:
157-172,
1972.
|
| 242. |
Frigyesi, T. L., and
R. Schwartz.
Cortical control of thalamic sensorimotor relay activities in the cat and the squirrel monkey. In:
Corticothalamic Projections and Sensorimotor Activities,
edited by T. Frigyesi,
E. Rinvik, and
M. D. Yahr.
New York:
Raven,
1972,
p. 161-191.
|
| 243. |
Frost, D. O., and
V. S. Caviness Jr.
Radial organization of thalamic projections to the neocortex in the mouse.
J. Comp. Neurol.
194:
369-393,
1980.
|
| 244. |
Fujita, Y., and
T. Sato.
Intracellular records from hippocampal pyramidal cells in rabbit during theta rhythm activity.
J. Neurophysiol.
27:
1011-1025,
1964.
|
| 245. |
Fukuda, Y., and
K. Iwama.
Inhibition des interneurones du corps genouillé latéral par activation de la formation réticulée.
Brain Res.
18:
548-551,
1970.
|
| 246. |
Fuller, C. A.,
R. Lydic,
F. M. Sulzman,
H. E. Albers,
B. Tepper, and
M. C. Moore‐Ede.
Circadian rhythm of body temperature persists after suprachiasmatic lesions in the squirrel monkey.
Am. J. Physiol.
241
(Regulatory Integrative Comp. Physiol. 10):
R385-R391,
1981.
|
| 247. |
Fuller, J. H.
Brain stem reticular units: some properties of the course and origin of the ascending trajectory.
Brain Res.
83:
349-367,
1975.
|
| 248. |
Fuller, J. H., and
J. D. Schlag.
Determination of antidromic excitation by the collision test: problems of interpretation.
Brain Res.
112:
283-298,
1976.
|
| 249. |
Fuster, J. M., and
A. A. Uyeda.
Facilitation of tachistoscopic performance by stimulation of midbrain tegmental points in the monkey.
Exp. Neurol.
6:
384-406,
1962.
|
| 250. |
Fuxe, K.,
T. Hökfelt, and
U. Ungestedt.
Morphological and functional aspects of central monoamine neurons.
Int. Rev. Neurobiol.
13:
93-126,
1970.
|
| 251. |
Galambos, R.,
G. Sheatz, and
V. G. Vernier.
Electrophysiological correlates of a conditioned response in cats.
Science
123:
376-377,
1956.
|
| 252. |
Gallagher, D. W., and
A. Pert.
Efferents to the brain stem nuclei (brain stem raphe, nucleus reticularis pontis caudalis and nucleus gigantocellularis) in the rat as demonstrated by microiontophoretically applied horseradish peroxidase.
Brain Res.
144:
257-275,
1978.
|
| 253. |
Garey, L. J., and
J. P. Hornung.
The use of ibotenic acid lesions for light and electron microscopic study of anterograde degeneration in the visual pathway of the cat.
Neurosci. Lett.
19:
117-123,
1980.
|
| 254. |
George, R.,
W. L. Haslett, and
D. J. Jenden.
A cholinergic mechanism in the pontine reticular formation: induction of paradoxical sleep.
Int. J. Neuropharmacol.
3:
541-552,
1964.
|
| 255. |
Giesler, G. J., Jr.,
R. P. Yezierski,
K. D. Gerhart, and
W. D. Willis.
Spinothalamic tract neurons that project to medial and/or lateral thalamic nuclei: evidence for a physiologically novel population of spinal cord neurons.
J. Neurophysiol.
46:
1285-1308,
1981.
|
| 256. |
Gillin, J. G.,
R. M. Post,
R. J. Wyatt,
F. K. Goodwin,
F. Snyder, and
W. Bunney Jr.
REM inhibitory effect of L‐dopa infusion during human sleep.
Electroencephalogr. Clin. Neurophysiol.
35:
181-186,
1973.
|
| 257. |
Glenn, L. L., and
W. C. Dement.
Membrane potential, synaptic activity, and excitability of hindlimb motoneurons during wakefulness and sleep.
J. Neurophysiol.
46:
839-854,
1981.
|
| 258. |
Glenn, L. L.,
J. Hada,
J. P. Roy,
M. Deschěnes, and
M. Steriade.
Anterograde tracer and field potential analysis of the neocortical layer I projection from nucleus ventralis medialis of the thalamus in cat.
Neuroscience
7:
1861-1877,
1982.
|
| 259. |
Glenn, L. L., and
M. Steriade.
Discharge rate and excitability of cortically projecting neurons in the intralaminar thalamic nuclei during waking and sleep states.
J. Neurosci.
2:
1387-1404,
1982.
|
| 260. |
Goebel, H. H.,
A. Komatsuzaki,
M. B. Bender, and
B. Cohen.
Lesions of the pontine tegmentum and conjugate gaze paralysis.
Arch. Neurol.
24:
431-440,
1971.
|
| 261. |
Goldberg, M. P.,
D. Riew,
E. Vivaldi,
R. W. McCarley, and
J. A. Hobson.
Activation of muscarinic receptors in the pontine tegmentum can trigger REM sleep: effects of bethanechol (Abstract).
Sleep Res.
10:
81,
1981.
|
| 262. |
Graham, J., and
N. Berman.
Origins of the pretectal and tectal projections to the central lateral nucleus in the cat.
Neurosci. Lett.
26:
209-214,
1981.
|
| 263. |
Grantyn, A., and
R. Grantyn.
Axonal patterns and sites of termination of cat superior colliculus neurons projecting in the tecto‐bulbo‐spinal tract.
Exp. Brain Res.
46:
243-256,
1982.
|
| 264. |
Grantyn, A.,
M. Mancia,
G. Broggi, and
M. Margnelli.
Intralaminar thalamic influences on bulbo‐pontine and mesencephalic neurones as revealed by intracellular recording.
Brain Res.
33:
223-226,
1971.
|
| 265. |
Grantyn, R.,
M. Margnelli,
M. Mancia, and
A. Grantyn.
Postsynaptic potentials in the mesencephalic and ponto‐medullary reticular regions underlying descending limbic influences.
Brain Res.
56:
107-121,
1973.
|
| 266. |
Graybiel, A. M.
Direct and indirect preoculomotor pathways of the brainstem: an autoradiographic study of the pontine reticular formation in the cat.
J. Comp. Neurol.
175:
37-78,
1977.
|
| 267. |
Graybiel, A. M., and
R. P. Elde.
Somatostatin‐like immunoreactivity characterizes neurons of the nucleus reticularis thalami in the cat and monkey.
J. Neurosci.
3:
1308-1321,
1983.
|
| 268. |
Graybiel, A. M., and
E. A. Hartwieg.
Some afferent connections of the oculomotor complex in the cat: an experimental study with tracer techniques.
Brain Res.
81:
543-551,
1974.
|
| 269. |
Green, J. D., and
A. Arduini.
Hippocampal electrical activity in arousal.
J. Neurophysiol.
17:
533-557,
1954.
|
| 270. |
Grofova, I.
The identification of striatal and pallidal neurons projecting to substantia nigra. An experimental study by means of retrograde axonal transport of horseradish peroxidase.
Brain Res.
91:
286-291,
1975.
|
| 271. |
Grofova, I.,
O. P. Ottersen, and
E. Rinvik.
Mesencephalic and diencephalic afferents to the superior colliculus and periaqueductal gray substance demonstrated by retrograde axonal transport of horseradish peroxidase in the cat.
Brain Res.
146:
205-220,
1978.
|
| 272. |
Grossman, S. P.,
D. Dacey,
A. E. Halaris,
T. Collier, and
A. Routtenberg.
Aphagia and adipsia after preferential destruction of nerve cell bodies in hypothalamus.
Science
202:
537-539,
1978.
|
| 273. |
Gücer, G.
The effect of sleep upon the transmission of afferent activity in the somatic afferent system.
Exp. Brain Res.
34:
287-298,
1979.
|
| 274. |
Guilleminault, C.,
R. L. Ariagno,
L. S. Forno,
L. Nagel,
R. Baldwin, and
M. Owen.
Obstructive sleep apnea and near miss for SIDS. 1. Report of an infant with sudden death.
Pediatrics
63:
837-843,
1979.
|
| 275. |
Guilleminault, C.,
R. Ariagno,
R. Korobkin,
L. Nagel,
R. Baldwin,
S. Coons, and
M. Owen.
Mixed and obstructive sleep apnea and near miss for sudden infant death syndrome. 2. Comparison of near miss and normal control infants by age.
Pediatrics
64:
882-891,
1979.
|
| 276. |
Guilleminault, C., and
W. C. Dement.
Sleep Apnea Syndromes.
New York:
Liss,
1978.
|
| 277. |
Guilleminault, C.,
F. L. Eldridge,
A. Tilkian,
F. B. Simmons, and
W. C. Dement.
Sleep apnea syndrome due to upper airway obstruction: a review of 25 cases.
Arch. Intern. Med.
137:
296-300,
1977.
|
| 278. |
Hajdu, F.,
G. Somogyi, and
T. Tömböl.
Neuronal and synaptic arrangement in the lateralis posterior‐pulvinar complex of the thalamus in the cat.
Brain Res.
73:
89-104,
1974.
|
| 279. |
Hammer, R. P., Jr.,
R. D. Lindsay, and
A. B. Scheibel.
Development of the brain stem reticular core: an assessment of dendritic state and configuration in the perinatal rat.
Dev. Brain Res.
7:
179-190,
1981.
|
| 280. |
Harding, B. N., and
T. P. S. Powell.
An electron microscopic study of the centre‐median and ventrolateral nuclei of the thalamus in the monkey.
Proc. R. Soc. London
279:
357-412,
1977.
|
| 281. |
Hartmann, E., and
T. J. Bridwell.
Effects of AMPT, L‐dopa and L‐tryptophan on sleep in the rat (Abstract).
Psychophysiology
7:
313,
1970.
|
| 282. |
Hartmann, E.,
T. J. Bridwell, and
J. J. Schildkraut.
α‐Methylparatyrosine and sleep in the rat.
Psychopharmacologia
21:
157-164,
1971.
|
| 283. |
Hartmann, E., and
G. Zwilling.
Effect of α‐ and β‐blockers on sleep patterns in the rat (Abstract).
Sleep Res.
1:
54,
1972.
|
| 284. |
Hartmann, E.,
G. Zwilling, and
S. List.
Effects of an alpha‐adrenergic blocker on sleep in the rat (Abstract).
Sleep Res.
2:
58,
1973.
|
| 285. |
Hartmann, E.,
G. Zwilling,
V. Pochay, and
R. Chung.
Intraventricular administration of neurotransmitters: effects on sleep (Abstract).
Sleep Res.
3:
41,
1974.
|
| 286. |
Hartse, K. M.,
S. F. Eisenhart,
B. M. Bergmann, and
A. Rechtschaffen.
Ventral hippocampus spikes during sleep, wakefulness and arousal in the cat.
Sleep
1:
231-246,
1979.
|
| 287. |
Hashikawa, T., and
K. Kawamura.
Identification of cells of origin of tectopontine fibers in the cat superior colliculus: an experimental study with the horseradish peroxidase method.
Brain Res.
130:
65-79,
1977.
|
| 288. |
Hattori, T.,
H. C. Fibiger, and
P. L. McGeer.
Demonstration of a pallido‐nigral projection innervating dopaminergic neurons.
J. Comp. Neurol.
162:
487-504,
1975.
|
| 289. |
Hazlett, J. C.,
C. R. Dutta, and
C. A. Fox.
The neurons in the centromedian‐parafascicular complex of the monkey (Macaco mulatta): a Golgi study.
J. Comp. Neurol.
168:
41-74,
1976.
|
| 290. |
Hazra, J.
Effect of hemicholinium‐3 on slow wave and paradoxical sleep of cat.
Eur. J. Pharmacol.
11:
395-397,
1970.
|
| 291. |
Hendry, S. H. C., and
E. G. Jones.
Sizes and distributions of intrinsic neurons incorporating tritiated GABA in monkey sensory‐motor cortex.
J. Neurosci.
1:
390-408,
1981.
|
| 292. |
Hendry, S. H. C.,
E. G. Jones, and
J. Graham.
Thalamic relay nuclei for cerebellar and certain related fiber systems in the cat.
J. Comp. Neurol.
185:
679-714,
1979.
|
| 293. |
Henley, K., and
A. R. Morrison.
A re‐evaluation of the effects of lesions of the pontine tegmentum and locus coeruleus on phenomena of paradoxical sleep in the cat.
Acta Neurobiol. Exp.
34:
215-232,
1974.
|
| 294. |
Henn, V.,
V. Büttner, and
J. Büttner‐Ennever.
Supranucleus organization of the oculomotor system—anatomical and physiological investigations. In:
Disorders of Ocular Motility: Neurophysiological and Clinical Aspects,
edited by G. Kommerell.
Munich, West Germany:
Bermann,
1978,
p. 129-143.
|
| 295. |
Henneman, E.,
G. Somjen, and
D. O. Carpenter.
Functional significance of cell size in spinal motoneurons.
J. Neurophysiol.
28:
560-580,
1965.
|
| 296. |
Henriksen, S. J.,
B. L. Jacobs, and
W. C. Dement.
Dependence of REM sleep PGO waves on cholinergic mechanisms.
Brain Res.
48:
412-416,
1972.
|
| 297. |
Henriksen, S.,
B. Jacobs,
W. Dement, and
J. Barchas.
Catecholamine mechanisms: their presumptive role in the generation of REM sleep PGO waves. In:
Frontiers in Catecholamine Research,
edited by E. Usdin and
S. H. Snyder.
Oxford, UK:
Pergamon,
1973,
p. 759-762.
|
| 298. |
Herkenham, M.
The afferent and efferent connections of the ventromedial thalamic nucleus in the rat.
J. Comp. Neurol.
183:
487-518,
1979.
|
| 299. |
Herkenham, M.
Laminar organization of thalamic projections to the rat neocortex.
Science
207:
532-535,
1980.
|
| 300. |
Herman, J. H.,
H. P. Roffwarg,
C. J. Rosenmann, and
E. J. Tauber.
Binocular depth perception following REM deprivation or awake state visual deprivation.
Psychophysiology
17:
236-242,
1980.
|
| 301. |
Herman, Z. S.
The effects of noradrenaline on rat's behavior.
Psychopharmacologia
16:
369-374,
1970.
|
| 302. |
Hernandez‐Peon, R.
A cholinergic hypnogenic limbic forebrain‐hindbrain circuit. In:
Neurophysiologie des états du sommeil,
edited by M. Jouvet.
Lyons, France:
CNRS,
1965,
p. 63-88.
|
| 303. |
Hersch, S. M., and
E. L. White.
Thalamocortical synapses with corticothalamic projection neurons in mouse SmI cortex: electron microscopic demonstration of a monosynaptic feedback loop.
Neurosci. Lett.
24:
207-210,
1981.
|
| 304. |
Higuchi, T.,
Y. Takahashi,
K. Takahashi,
Y. Niimi, and
A. Miyasita.
Twenty‐four‐hour secretory patterns of growth hormones, prolactin, and Cortisol in narcolepsy.
J. Clin. Endocrinol. Metab.
49:
197-204,
1979.
|
| 305. |
Hirsch, J. C.,
A. Fourment, and
M. E. Marc.
Electrophysiological study of the peripeniculate region during natural sleep of the cat.
Exp. Neurol.
2:
436-454,
1982.
|
| 306. |
Hirsch, J. C.,
A. Fourment, and
M. E. Marc.
Sleep‐related variations of membrane potential in the lateral geniculate body relay neurons of the cat.
Brain Res.
259:
308-312,
1983.
|
| 307. |
Hishikawa, T.,
K. Nakai,
H. Ida, and
Z. Kaneko.
The effect of imipramine, desmethylimipramine and chlorpromazine on the sleep‐wakefulness cycle of the cat.
Electroencephalogr. Clin. Neurophysiol.
19:
518-521,
1965.
|
| 308. |
Hobson, J. A.
The effect of chronic brain stem lesions on cortical and muscular activity during sleep and waking in the cat.
Electroencephalogr. Clin. Neurophysiol.
19:
41-62,
1965.
|
| 309. |
Hobson, J. A.
Sleep after exercise.
Science
162:
1503-1505,
1968.
|
| 310. |
Hobson, J. A.
The cellular basis of sleep cycle control. In:
Advances in Sleep Research,
edited by E. D. Weitzman.
New York:
Spectrum,
1974,
vol. 1,
p. 217-250.
|
| 311. |
Hobson, J. A.
Dreaming sleep attacks and desynchronized sleep enhancement.
Arch. Gen. Psychiatry
32:
1421-1424,
1975.
|
| 312. |
Hobson, J. A.
The sleep‐dream cycle: a neurobiological rhythm. In:
Pathobiology Annual,
edited by H. L. Ioachim.
New York:
Appleton‐Century‐Crofts,
1975,
p. 369-703.
|
| 313. |
Hobson, J. A.
The Ethology of Sleep.
Manati, Puerto Rico:
Roche,
1979.
|
| 314. |
Hobson, J. A., and
M. A. B. Brazier (editors).
The Reticular Formation Revisited.
New York:
Raven,
1979.
|
| 315. |
Hobson, J. A.,
F. Goldfrank, and
F. Snyder.
Respiration and mental activity in sleep.
J. Psychiatr. Res.
3:
79-90,
1965.
|
| 316. |
Hobson, J. A.,
R. W. McCarley,
R. Freedman, and
R. T. Pivik.
Time course of discharge rate changes by cat pontine brain stem neurons during sleep cycle.
J. Neurophysiol.
37:
1297-1309,
1974.
|
| 317. |
Hobson, J. A.,
R. W. McCarley, and
J. P. Nelson.
Location and spike‐train characteristics of cells in anterodorsal pons having selective decreases in discharge rates during desynchronized sleep.
J. Neurophysiol.
50:
770-783,
1983.
|
| 318. |
Hobson, J. A.,
R. W. McCarley,
R. T. Pivik, and
R. Freedman.
Selective firing by cat pontine brain stem neurons in desynchronized sleep.
J. Neurophysiol.
37:
497-511,
1974.
|
| 319. |
Hobson, J. A.,
R. W. McCarley, and
P. W. Wyzinski.
Sleep cycle oscillation: reciprocal discharge by two brainstem neuronal groups.
Science
189:
55-58,
1975.
|
| 320. |
Hobson, J. A., and
A. B. Scheibel.
The brainstem core: sensorimotor integration and behavioral state control.
Neurosci. Res. Program Bull.
18:
1-173,
1980.
|
| 321. |
Hobson, J. A.,
T. Spagna, and
R. Malenka.
Ethology of sleep studied with time‐lapse photography: postural immobility and sleep‐cycle phase in humans.
Science
201:
1251-1253,
1978.
|
| 322. |
Hoffer, B. J.,
G. R. Siggins,
A. P. Oliver, and
F. E. Bloom.
Activation of the pathway from locus coeruleus to rat cerebellar Purkinje neurons: pharmacological evidence of noradrenergic central inhibition.
J. Pharmacol. Exp. Ther.
184:
553-569,
1973.
|
| 323. |
Hoffer, B. J.,
G. R. Siggins,
D. J. Woodward, and
F. E. Bloom.
Spontaneous discharge of Purkinje neurons after destruction of catecholamine‐containing afferents by 6‐hydroxydopamine.
Brain Res.
30:
425-430,
1971.
|
| 324. |
Hoffman, J. S., and
E. Domino.
Comparative effects of reserpine on the sleep cycle of man and cat.
J. Pharmacol. Exp. Ther.
170:
190-198,
1969.
|
| 325. |
Holcombe, V., and
W. C. Hall.
The laminar origin and distribution of the crossed tectoreticular pathways.
J. Neurosci.
1:
1103-1112,
1981.
|
| 326. |
Horvath, F. E., and
P. Buser.
Thalamo‐caudate‐cortical relationships in synchronized activity. I. Differentiation between ventral and dorsal spindle systems.
Brain Res.
39:
21-41,
1972.
|
| 327. |
Hoshino, K., and
O. Pompeiano.
Selective discharge of pontine neurons during the postural atonia produced by an anticholinesterase in the decerebrate cat.
Arch. Ital. Biol.
114:
244-247,
1976.
|
| 328. |
Hosoya, Y., and
M. Matsushita.
Brainstem projections from the lateral hypothalamic area in the rat, as studied with autoradiography.
Neurosci. Lett.
24:
111-116,
1981.
|
| 329. |
Houser, C. R.,
J. E. Vaughn,
R. P. Barber, and
E. Roberts.
GABA neurons are the major cell type of the nucleus reticularis thalami.
Brain Res.
200:
341-354,
1980.
|
| 330. |
Hubbard, J. E., and
V. DiCarlo.
Fluorescence histochemistry of monoamine‐containing cell bodies in the brain stem of the squirrel monkey (Saimiri sciureus).
J. Comp. Neurol.
147:
553-566,
1973.
|
| 331. |
Hubel, D. H.
Single unit activity in striate cortex of unrestrained cats.
J. Physiol. London
147:
226-238,
1959.
|
| 332. |
Hubel, D. H.
Single unit activity in lateral geniculate body and optic tract of unrestrained cats.
J. Physiol. London
150:
91-104,
1960.
|
| 333. |
Hugelin, A.
Does the respiratory rhythm originate from a reticular oscillator in the waking state? In:
The Reticular Formation Revisited,
edited by J. A. Hobson and
M. A. B. Brazier.
New York:
Raven,
1979,
p. 261-274.
|
| 334. |
Huttenlocher, P. R.
Evoked and spontaneous activity in single units of medial brain stem during natural sleep and waking.
J. Neurophysiol.
24:
451-468,
1961.
|
| 335. |
Ibuka, N.,
S. T. Inouye, and
H. Hawamura.
Analysis of sleep‐wakefulness rhythms in male rats after suprachiasmatic nucleus lesions and ocular enucleations.
Brain Res.
122:
33-47,
1977.
|
| 336. |
Inouye, S. T., and
H. Kawamura.
Persistence of circadian rhythmicity in a mammalian hypothalamic “island” containing the suprachiasmatic nucleus.
Proc. Natl. Acad. Sci. USA
76:
5962-5966,
1979.
|
| 337. |
Inubushi, S.,
T. Kobayashi,
T. Oshima, and
S. Torii.
Intracellular recordings from the motor cortex during EEG arousal in unanesthetized brain preparations of the cat.
Jpn. J. Physiol.
28:
669-688,
1978.
|
| 338. |
Inubushi, S.,
T. Kobayashi,
T. Oshima, and
S. Torii.
An intracellular analysis of EEG arousal in cat motor cortex.
Jpn. J. Physiol.
28:
689-708,
1978.
|
| 339. |
Iskander, T. N., and
R. Kaebling.
Catecholamine, a dream sleep model and depression.
Am. J. Psychiatry
127:
43-50,
1970.
|
| 340. |
Ito, M.,
M. Udo, and
N. Mano.
Long inhibitory and excitatory pathways converging onto cat reticular and Deiters' neurons and their relevance to reticulofugal axons.
J. Neurophysiol.
33:
210-226,
1970.
|
| 341. |
Itoh, K., and
N. Mizuno.
Direct projections from the mesodiencephalic midline areas to the pericruciate cortex in the cat: an experimental study with the horseradish peroxidase method.
Brain Res.
116:
492-497,
1976.
|
| 342. |
Itoh, K., and
N. Mizuno.
Topographical arrangement of thalamocortical neurons in the centrolateral nucleus (CL) of the cat, with special reference to a spino‐thalamo‐motor cortical path through the CL.
Exp. Brain Res.
30:
471-480,
1977.
|
| 343. |
Jacobs, B. L.,
R. Asher,
S. J. Henriksen, and
W. C. Dement.
Electroencephalographic and behavioural effects of stimulation of the raphe nuclei in cats (Abstract).
Sleep Res.
1:
23,
1972.
|
| 344. |
Jacobs, B. L., and
B. E. Jones.
The role of central monoamine and acetylcholine systems in sleep‐wakefulness states: mediation or modulation. In:
Cholinergic‐Monoaminergic Interactions in the Brain,
edited by L. L. Butcher.
New York:
Academic,
1978,
p. 271-290.
|
| 345. |
Jahnsen, H., and
R. Llinás.
Electrophysiological properties of guinea‐pig thalamic neurones: an in vitro study.
J. Physiol. London
349:
205-226,
1984.
|
| 346. |
Jahnsen, H., and
R. Llinás.
Ionic basis for the electroresponsiveness and oscillatory properties of guinea‐pig thalamic neurones in vitro.
J. Physiol. London
349:
227-247,
1984.
|
| 347. |
Jalfre, M.,
M. A. Monachon, and
W. Haefely.
Drug and PGO‐waves in the cat. In:
Proc. Can. Int. Symp. Sleep, 1st,
1972,
p. 155-184.
|
| 348. |
Jalfre, M.,
M. A. Ruch‐Monachon, and
W. Haefely.
Methods for assessing the interaction of agents with 5‐hydroxytryptamine neurons and receptors in the brain.
Adv. Biochem. Psychopharmacol.
10:
121-134,
1974.
|
| 349. |
Jankowska, E.,
S. Lund,
A. Lundberg, and
O. Pompeiano.
Inhibitory effects evoked through ventral reticulospinal pathways.
Arch. Ital. Biol.
106:
124-140,
1968.
|
| 350. |
Jasper, H. H.
Diffuse projection systems: the integrative action of the thalamic reticular system.
Electroencephalogr. Clin. Neurophysiol.
1:
405-420,
1949.
|
| 351. |
Jasper, H. H.
Recent advances in our understanding of ascending activities of the reticular system. In:
Reticular Formation of the Brain,
edited by H. H. Jasper,
L. D. Proctor,
R. S. Knighton,
W. C. Noshay, and
R. T. Costello.
Boston, MA:
Little, Brown,
1958,
p. 319-331.
|
| 352. |
Jasper, H. H.
Unspecific thalamocortical relations. In:
Handbook of Physiology. Neurophysiology,
edited by J. Field and
H. W. Magoun.
Washington, DC:
Am. Physiol. Soc.,
1960,
sect. 1,
vol. II,
chapt. LIII,
p. 1307-1321.
|
| 353. |
Jasper, H. H.
Chairman's summary. In:
Corticothalamic Projections and Sensorimotor Activities,
edited by T. Frigyesi,
E. Rinvik, and
M. D. Yahr.
New York:
Raven,
1972,
p. 305-308.
|
| 354. |
Jasper, H. H.
Problems of relating cellular or modular specificity to cognitive functions: importance of state‐dependent reaction. In:
The Organization of the Cerebral Cortex,
edited by F. O. Schmitt,
F. G. Worden,
G. Adelman, and
S. G. Dennis.
Cambridge, MA:
MIT Press,
1981,
p. 375-393.
|
| 355. |
Jasper, H. H., and
I. Koyama.
Rate of release of aminoacids from the cerebral cortex in the cat as affected by brainstem and thalamic stimulation.
Can. J. Physiol. Pharmacol.
47:
889-905,
1969.
|
| 356. |
Jasper, H.,
R. Naquet, and
E. E. King.
Thalamocortical recruiting responses in sensory receiving areas in the cat.
Electroencephalogr. Clin. Neurophysiol.
7:
99-114,
1955.
|
| 357. |
Jasper, H.,
G. F. Rici, and
B. Doane.
Patterns of cortical neuron discharge during conditioned responses in monkeys. In:
Neurological Basis of Behaviour,
edited by G. E. W. Wolstenholme and
M. J. O'Connor.
Boston, MA:
Little, Brown,
1958,
p. 277-294.
(Ciba Found. Symp.)
|
| 358. |
Jasper, H. H., and
J. Tessier.
Acetylcholine liberation from cerebral cortex during paradoxical (REM) sleep.
Science
172:
601-602,
1971.
|
| 359. |
Jeannerod, M., and
S. Kiyono.
Décharge unitaire de la formation réticulée pontique et activité phasique ponto‐géniculo‐occipitale chez le chat sous réserpine.
Brain Res.
12:
112-128,
1969.
|
| 360. |
Jewett, R. E., and
S. Norton.
Effects of some stimulant and depressant drugs on sleep cycles of cats.
Exp. Neurol.
15:
463-474,
1966.
|
| 361. |
Jinnai, K., and
Y. Matsuda.
Thalamocaudate projection neurons with a branching axon to the cerebral motor cortex.
Neurosci. Lett.
26:
95-99,
1981.
|
| 362. |
Jones, B. E.
The respective involvement of noradrenaline and its deaminated metabolites in waking and paradoxical sleep: a neuropharmacological model.
Brain Res.
39:
121-136,
1972.
|
| 363. |
Jones, B. E.,
S. T. Harper, and
A. E. Halaris.
Effects of locus coeruleus lesions upon cerebral monoamine content, sleep‐wakefulness states and the response to amphetamine in the cat.
Brain Res.
124:
473-496,
1977.
|
| 364. |
Jones, B. E., and
R. Y. Moore.
Catecholamine‐containing neurons of the nucleus locus coeruleus in the cat.
J. Comp. Neurol.
157:
42-51,
1974.
|
| 365. |
Jones, B. E., and
R. Y. Moore.
Ascending projections of the locus coeruleus in the rat. II. Autoradiographic study.
Brain Res.
127:
23-53,
1977.
|
| 366. |
Jones, E. G.
Possible determinants of the degree of retrograde neuronal labeling with horseradish peroxidase.
Brain Res.
85:
249-253,
1975.
|
| 367. |
Jones, E. G.
Some aspects of the organization of the thalamic reticular complex.
J. Comp. Neurol.
162:
285-308,
1975.
|
| 368. |
Jones, E. G.
Varieties and distribution of non‐pyramidal cells in the somatic sensory cortex of the squirrel monkey.
J. Comp. Neurol.
160:
205-267,
1975.
|
| 369. |
Jones, E. G.
Anatomy of cerebral cortex: columnar input‐ouput organization. In:
The Organization of the Cerebral Cortex,
edited by F. O. Schmitt,
F. G. Worden,
G. Adelman, and
S. G. Dennis.
Cambridge, MA:
MIT Press,
1981,
p. 199-235.
|
| 370. |
Jones, E. G.
Functional subdivision and synaptic organization of the mammalian thalamus. In:
Neurophysiology IV,
edited by R. Porter.
Baltimore, MD:
University Park,
1981,
vol. 25,
p. 173-245.
(Int. Rev. Physiol. Ser.)
|
| 371. |
Jones, E. G., and
H. Burton.
Cytoarchitecture and somatic sensory connectivity of thalamic nuclei other than the ventrobasal complex in the cat.
J. Comp. Neurol.
154:
395-432,
1974.
|
| 372. |
Jones, E. G.,
H. Burton,
C. B. Saper, and
L. W. Swanson.
Midbrain, diencephalic and cortical relationships of the basal nucleus of Meynert and associated structures in primates.
J. Comp. Neurol.
167:
385-420,
1976.
|
| 373. |
Jones, E. G., and
R. Y. Leavitt.
Retrograde axonal transport and the demonstration of nonspecific projections to the cerebral cortex and striatum from thalamic intralaminar nuclei in the rat, cat and monkey.
J. Comp. Neurol.
154:
349-378,
1974.
|
| 374. |
Jouvet, M.
Recherches sur les structures nerveuses et les mécanismes responsables des différentes phases du sommeil physiologique.
Arch. Ital. Biol.
100:
125-206,
1962.
|
| 375. |
Jouvet, M.
Mechanisms of the states of sleep: a neuropharmacological approach. In:
Sleep and Altered States of Consciousness,
edited by S. S. Kety.
Baltimore, MD:
Williams & Wilkins,
1967,
p. 86-126.
|
| 376. |
Jouvet, M.
Neurophysiology of the states of sleep. In:
The Neurosciences: A Study Program,
edited by G. C. Quarton,
T. Melnechuk, and
F. O. Schmitt.
New York:
Rockefeller Univ. Press,
1967,
p. 529-544.
|
| 377. |
Jouvet, M.
Biogenic amines and the states of sleep.
Science
163:
32-41,
1969.
|
| 378. |
Jouvet, M.
The role of monoamines and acetylcholine‐containing neurons in the regulation of the sleep‐waking cycle.
Ergeb. Physiol. Biol. Chem. Exp. Pharmakol.
64:
166-307,
1972.
|
| 379. |
Jouvet, M.
Essai sur le réve.
Arch. Ital. Biol.
111:
564-576,
1973.
|
| 380. |
Jouvet, M., and
J. F. Delorme.
Locus coeruleus et sommeil paradoxal.
C. R. Soc. Biol.
159:
895-899,
1965.
|
| 381. |
Jouvet, M.,
M. Jeannerod, and
F. Delorme.
Organisation du système responsable de l'activité phasique au cours du sommeil paradoxal.
C. R. Soc. Biol.
7:
1599-1604,
1965.
|
| 382. |
Jouvet, M., and
F. Michel.
Correlations électromyographiques du sommeil chez le chat décortiqué et mésencéphalique chronique.
C. R. Soc. Biol.
153:
422-425,
1959.
|
| 383. |
Kaelber, W. W., and
T. B. Smith.
Projections of the zona incerta in the cat, with stimulation controls.
Exp. Neurol.
63:
177-200,
1979.
|
| 384. |
Kales, A.,
R. Cadieux,
C. R. Soldatos, and
T. L. Tan.
Successful treatment of narcolepsy with propanolol: a case report.
Arch. Neurol.
36:
650-651,
1979.
|
| 385. |
Kales, A.,
A. Jacobson,
M. J. Paulson,
J. Kales, and
R. D. Walter.
Somnambulism: psychophysiological correlates. I. All‐night EEG studies.
Arch. Gen. Psychiatry
14:
586-594,
1966.
|
| 386. |
Kales, A., and
J. D. Kales.
Recent findings in the diagnosis and treatment of disturbed sleep.
N. Engl. J. Med.
290:
487-499,
1974.
|
| 387. |
Kales, A.,
C. R. Solidatos,
R. Cadieux,
E. O. Bixler,
T. L. Tan, and
M. B. Scharf.
Propranolol in the treatment of narcolepsy.
Ann. Intern. Med.
91:
741-743,
1979.
|
| 388. |
Kanamori, N.,
K. Sakai, and
M. Jouvet.
Neuronal activity specific to paradoxical sleep in the ventromedial medullary reticular formation of unrestrained cats.
Brain Res.
189:
251-255,
1980.
|
| 389. |
Kandel, E. R.
Cellular Basis of Behavior: An Introduction to Behavioral Neurobiology.
San Francisco, CA:
Freeman,
1976.
|
| 390. |
Kasamatsu, T.
Maintained and evoked unit activity in the mesencephalic reticular formation of the freely behaving cat.
Exp. Neurol.
28:
450-470,
1970.
|
| 391. |
Kasamatsu, T., and
P. Heggelund.
Single cell responses in cat visual cortex to visual stimulation during iontophoresis of noradrenaline.
Exp. Brain Res.
45:
317-327,
1982.
|
| 392. |
Kasamatsu, T., and
J. D. Pettigrew.
Depletion of brain catecholamines: failure of ocular dominance shift after monocular occlusion in kittens.
Science
194:
206-208,
1976.
|
| 393. |
Katayama, Y.,
Y. Ueno,
T. Tsukiyama, and
T. Tsubokawa.
Long lasting suppression of firing of cortical neurons and decrease in cortical blood flow following train pulse stimulation of the locus coeruleus in the cat.
Brain Res.
216:
173-179,
1981.
|
| 394. |
Keller, E. L.
Participation of medial pontine reticular formation in eye movement generation in monkey.
J. Neurophysiol.
37:
316-332,
1974.
|
| 395. |
Kennedy, H., and
C. Balaydier.
Direct projections from thalamic intralaminar nuclei to extra‐striate visual cortex in the cat traced with horseradish peroxidase.
Exp. Brain Res.
28:
133-139,
1977.
|
| 396. |
Kernell, D., and
B. Zwaagstra.
Input conductance, axonal conduction velocity and cell size among hindlimb motoneurones of the cat.
Brain Res.
204:
311-326,
1981.
|
| 397. |
Kevetter, G. A., and
W. D. Willis.
Spinothalamic cells in the rat lumbar cord with collaterals to the medullary reticular formation.
Brain Res.
238:
181-185,
1982.
|
| 398. |
Khazan, N., and
P. Brown.
Differential effects of three tricyclic antidepressants on sleep and REM sleep in the rat.
Life Sci.
9:
279-284,
1970.
|
| 399. |
Khazan, N., and
C. H. Sawyer.
Mechanisms of paradoxical sleep as revealed by neurophysiology and pharmacologic approaches in the rabbit.
Psychopharmacologia
5:
457-462,
1964.
|
| 400. |
Kievit, J., and
H. G. J. M. Kuypers.
Fastigial cerebellar projections to the ventrolateral nucleus of the thalamus and the organization of the descending pathways. In:
Corticothalamic Projections and Sensorimotor Activities,
edited by T. Frigyesi,
E. Rinvik, and
M. D. Yahr.
New York:
Raven,
1972,
p. 91-111.
|
| 401. |
Kievet, J., and
H. G. J. M. Kuypers.
Basal forebrain and hypothalamic connections to the frontal and parietal cortex in the rhesus monkey.
Science
187:
660-662,
1975.
|
| 402. |
Killackey, H. P., and
F. F. Ebner.
Two different types of thalamocortical projections to a single cortical area in mammals.
Brain Behav. Evol.
6:
141-169,
1972.
|
| 403. |
Kimura, H.,
P. L. McGeer,
J. H. Peng, and
E. G. McGeer.
The central cholinergic system studied by choline acetyltransferase immunihistochemistry in the cat.
J. Comp. Neurol.
200:
151-201,
1981.
|
| 404. |
King, A. B., and
S. M. Robinson.
Ventilation response to hypoxia and acute mountain sickness.
Aerosp. Med.
43:
419-421,
1972.
|
| 405. |
King, C. D.
The pharmacology of rapid eye movement sleep.
Adv. Pharmacol. Chemother.
9:
1-91,
1971.
|
| 406. |
King, C. D., and
E. J. Jewett.
The effects of γ‐methyltyrosine on sleep and brain norepinephrine in cats.
J. Pharmacol. Exp. Ther.
177:
188-195,
1971.
|
| 407. |
Kitsikis, A., and
M. Steriade.
Thalamic, callosal and reticular converging inputs to parietal association cortex in cat.
Brain Res.
93:
516-524,
1975.
|
| 408. |
Kitsikis, A., and
M. Steriade.
Immediate behavioral effects of kainic acid injections into the midbrain reticular core.
Behav. Brain Res.
3:
361-380,
1981.
|
| 409. |
Klee, M. R.,
H. D. Lux, and
K. Offenloch.
Veränderungen der Membranpolarisation und der Erregbarkeit von Zellen der motorischen Rinde während hochfrequenter Reizung der Formation Reticularis.
Arch. Psychol. Gesamte Neurol.
205:
237-261,
1974.
|
| 410. |
Kleitman, N.
Sleep and Wakefulness.
Chicago, IL:
Univ. of Chicago Press,
1963.
|
| 411. |
Knight, D. P.
Histochemical demonstration of catecholamines and acetylcholinesterase in the same cell bodies in the locus coeruleus (rat hindbrain).
Proc. R. Microsc. Soc.
6:
26-27,
1970.
|
| 412. |
Kornhuber, H. H., and
L. Deecke.
Hirnpotentialànderungen bei Willkürbewegungen und passiven Bewegunger des Menschen: Bereitschaftspotential und reafferente Potentiale.
Pfluegers Arch. Gesamte Physiol. Menschen Tiere
284:
1-17,
1965.
|
| 413. |
Kostowski, W.,
E. Giacalone,
S. Garattini, and
L. Valzelli.
Electrical stimulation of midbrain raphe: biochemical, behavioural and bioelectrical effects.
Eur. J. Pharmacol.
7:
170-175,
1969.
|
| 414. |
Krammer, E. B.,
M. F. Lischka,
M. Karobath, and
G. Schonbeck.
Is there a selectivity of neuronal degeneration induced by intrastriatal injection of kainic acid?
Brain Res.
177:
577-582,
1979.
|
| 415. |
Kreindler, A., and
M. Steriade.
EEG patterns of arousal and sleep induced by stimulating various amygdaloid levels in the cat.
Arch. Ital. Biol.
102:
576-586,
1964.
|
| 416. |
Krettek, J. E., and
J. L. Price.
The cortical projections of the mediodorsal nucleus and adjacent thalamic nuclei in the rat.
J. Comp. Neurol.
171:
157-192,
1977.
|
| 417. |
Krnjević, K.
Chemical nature of synaptic transmission in vertebrates.
Physiol. Rev.
54:
418-540,
1974.
|
| 418. |
Krnjević, K.
Transmitters in motor systems. In:
Handbook of Physiology. The Nervous System. Motor Control,
edited by J. M. Brookhart and
V. B. Mountcastle.
Bethesda, MD:
Am. Physiol. Soc.,
1981,
sect. 1,
vol. II,
pt. 1,
chapt. 4,
p. 107-154.
|
| 419. |
Krnjević, K., and
J. W. Phillis.
Acetylcholine‐sensitive cells in the cerebral cortex.
J. Physiol. London
166:
296-327,
1963.
|
| 420. |
Krnjević, K.,
E. Puil, and
R. Werman.
EGTA and motoneuronal after‐potentials.
J. Physiol. London
275:
199-223,
1978.
|
| 421. |
Krnjević, K.,
R. Pumain, and
L. Renaud.
The mechanism of excitation by acetylcholine in the cerebral cortex.
J. Physiol. London
215:
247-268,
1971.
|
| 422. |
Krnjević, K., and
W. Reinhardt.
Choline excites cortical neurons.
Science
206:
1321-1323,
1979.
|
| 423. |
Krnjević, K., and
S. Schwartz.
Some properties of unresponsive cells in the cerebral cortex.
Exp. Brain Res.
3:
306-319,
1967.
|
| 424. |
Kromer, L. F., and
R. Y. Moore.
A study of the organization of the locus coeruleus projections to the lateral geniculate nuclei in the albino rat.
Neuroscience
5:
255-271,
1980.
|
| 425. |
Kucera, P., and
P. Favrod.
Suprachiasmatic nucleus projection to mesencephalic central gray in the woodmouse (Apodemus sylvaticus L.).
Neuroscience
4:
1705-1715,
1979.
|
| 426. |
Kuhar, M. J.,
G. K. Aghajanian, and
R. H. Roth.
Tryptophan hydroxylase activity and synaptosomal uptake of serotonin in discrete brain regions after midbrain raphe lesions: correlations with serotonin levels and histochemical fluorescence.
Brain Res.
44:
165-176,
1972.
|
| 427. |
Kunze, W.,
J. S. McKenzie, and
A. P. Bendrups.
An electrophysiological study of thalamo‐caudate neurones in the cat.
Exp. Brain Res.
36:
233-244,
1979.
|
| 428. |
Kupfer, D. J., and
M. B. Bowers Jr.
REM sleep and central monoamine oxidase inhibition.
Psychopharmacologia
27:
183-190,
1972.
|
| 429. |
Kurtz, D.,
J. Krieger, and
J. C. Stierle.
EMG activity of cricothyroid and chin muscles during wakefulness and sleeping in the sleep apnea syndrome.
Electroencephalogr. Clin. Neurophysiol.
45:
777-784,
1978.
|
| 430. |
Kuypers, H. G. J. M.
Cortico‐bulbar connexcions to the pons and lower brain stem in man. An anatomical study.
Brain
81:
364-388,
1958.
|
| 431. |
Kuypers, H. G. J. M., and
V. A. Maisky.
Retrograde axonal transport of horseradish peroxidase from spinal cord to brain stem cell group in the cat.
Neurosci. Lett.
1:
9-14,
1975.
|
| 432. |
Laffont, F.,
A. Autret,
M. Minz,
T. Beillevaire,
H. P. Cathela, and
P. Castaigne.
Sleep respiratory arrhythmias in control subjects, narcoleptics and non‐cataplectic hypersomniacs.
Electroencephalogr. Clin. Neurophysiol.
44:
697-705,
1978.
|
| 433. |
Lamarre, Y.,
M. Filion, and
J. P. Cordeau.
Neuronal discharges of the ventrolateral nucleus of the thalamus during sleep and wakefulness in the cat. I. Spontaneous activity.
Exp. Brain Res.
12:
480-498,
1971.
|
| 434. |
Lamour, Y.,
P. Dutar, and
A. Jobert.
Excitatory effect of acetylcholine on different types of neurons in the first somatosensory neocortex of the rat: laminar distribution and pharmacological characteristics.
Neuroscience
7:
1483-1494,
1982.
|
| 435. |
Laurent, J. P., and
F. Ayalaguerrero.
Reversible suppression of ponto‐geniculo‐occipital waves by localized cooling during paradoxical sleep in cat.
Exp. Neurol.
49:
356-369,
1975.
|
| 436. |
Laurent, J. P.,
R. Cespuglio, and
M. Jouvet.
Delimitation des voies ascendantes de l'activité ponto‐geniculo‐occipitale chez le chat.
Brain Res.
65:
29-52,
1974.
|
| 437. |
Leger, L.,
L. Wiklund,
L. Descarries, and
M. Persson.
Description of an indolaminergic cell component in the cat locus coeruleus: a fluorescence histochemical and radioautographic study.
Brain Res.
168:
43-56,
1979.
|
| 438. |
Le Gros Clark, W. E.
The structure and connections of the thalamus.
Brain
55:
406-470,
1932.
|
| 439. |
Lehmann, J.,
J. I. Nagy,
S. Atmadja, and
H. C. Fibiger.
The nucleus basalis magnocellularis: the origin of a cholinergic projection to the neocortex of the rat.
Neuroscience
5:
1161-1174,
1980.
|
| 440. |
Leontovitch, T. A., and
G. P. Zhukova.
The specificity of the neuronal structure and topography of the reticular formation in the brain and spinal cord of carnivora.
J. Comp. Neurol.
121:
347-389,
1963.
|
| 441. |
Libet, B.,
H. Kobayashi, and
T. Tanaka.
Synaptic coupling into the production and storage of a neuronal memory trace.
Nature London
258:
155-157,
1975.
|
| 442. |
Lidbrink, P.,
G. Jonsson, and
K. Fuxe.
The effect of imipramine‐like drugs and antihistamine drugs on uptake mechanisms in the central noradrenaline and 5‐hydroxytryptamine neurons.
Neuropharmacology
10:
521-536,
1971.
|
| 443. |
Lidov, H. G. W.,
R. Grzanna, and
M. E. Molliver.
The serotonin innervation of the cerebral cortex in the rat—an immunohistochemical analysis.
Neuroscience
5:
207-227,
1980.
|
| 444. |
Lindsley, D. B.,
J. W. Bowden, and
H. W. Magoun.
Effect upon the EEG of acute injury to the brain stem activating system.
Electroencephalogr. Clin. Neurophysiol.
1:
475-486,
1949.
|
| 445. |
Lindsley, D. B.,
L. H. Schreiner,
W. B. Knowles, and
H. W. Magoun.
Behavioral and EEG changes following chronic brain stem lesions in the cat.
Electroencephalogr. Clin. Neurophysiol.
2:
483-498,
1950.
|
| 446. |
Lindstrom, S.
Synaptic organization of inhibitory pathways to principal cells in the lateral geniculate nucleus of the cat.
Brain Res.
234:
447-453,
1982.
|
| 447. |
Lindvall, O., and
A. Björklund.
The organization of the ascending catecholamine neuron systems in the rat brain as revealed by the glyoxylic acid fluorescence method.
Acta Physiol. Scand. Suppl.
412:
1-48,
1974.
|
| 448. |
Lindvall, O.,
A. Björklund,
R. Y. Moore, and
U. Stenevi.
Mesencephalic dopamine neurons projecting to neocortex.
Brain Res.
81:
325-331,
1974.
|
| 449. |
Lindvall, O.,
A. Björklund,
A. Nobin, and
U. Stenevi.
The adrenergic innervation of the rat thalamus as revealed by the glyoxylic acid fluorescence method.
J. Comp. Neurol.
154:
317-348,
1974.
|
| 450. |
Lipski, J.
Antidromic activation of neurones as an analytical tool in the study of the central nervous system.
J. Neurosci. Methods
4:
1-32,
1981.
|
| 451. |
Livingston, M. S., and
D. H. Hubel.
Effects of sleep and arousal on the processing of visual information in the cat.
Nature London
291:
554-561,
1981.
|
| 452. |
Ljungberg, T., and
U. Ungerstedt.
Sensory inattention produced by 6‐hydroxydopamine‐induced degeneration of ascending dopamine neurons in the brain.
Exp. Neurol.
53:
585-600,
1976.
|
| 453. |
Llinás, R., and
H. Jahnsen.
Electrophysiology of mammalian thalamic neurones in vitro.
Nature London
297:
406-408,
1982.
|
| 454. |
Llinás, R., and
Y. Yarom.
Electrophysiology of mammalian inferior olivary neurones in vitro. Different types of voltage‐dependent ionic conductance.
J. Physiol. London
315:
549-567,
1981.
|
| 455. |
Llinás, R., and
Y. Yarom.
Properties and distribution of ionic conductances generating electroresponsiveness of mammalian inferior olivary neurones in vitro.
J. Physiol. London
315:
569-584,
1981.
|
| 456. |
Lorente De Nó, R.
Cerebral cortex: architecture, intracortical connections, motor projections. In:
Physiology of the Nervous System,
edited by J. F. Fulton.
New York:
Oxford Univ. Press,
1938,
p. 291-340.
|
| 457. |
Lotka, A.
Elements of Physical Biology.
New York:
Dover,
1956.
|
| 458. |
Lucero, M. A.
Lengthening of REM sleep duration consecutive to learning in the rat.
Brain Res.
20:
319-322,
1970.
|
| 459. |
Lynch, J. C.,
V. B. Mountcastle,
W. H. Talbot, and
T. C. T. Yin.
Parietal lobe mechanisms for directed visual attention.
J. Neurophysiol.
40:
362-389,
1977.
|
| 460. |
Macchi, G.,
M. Bentivoglio,
C. D'Atena,
P. Rossini, and
E. Tempesta.
The cortical projection of the thalamic intra‐laminar nuclei restudied by means of the HRP retrograde axonal transport.
Neurosci. Lett.
4:
121-126,
1977.
|
| 461. |
Macchi, G.,
M. Bentivoglio,
M. Molinari, and
D. Minciacchi.
The thalamo‐caudate versus thalamo‐cortical projections as studied in the cat with fluorescent retrograde double labeling.
Exp. Brain Res.
54:
225-239,
1984.
|
| 462. |
MacGregor, R. J.,
R. Prieto‐Diaz,
S. W. Miller, and
P. M. Groves.
Statistical properties of neurons in the rat mesencephalic reticular formation.
Brain Res.
64:
167-187,
1973.
|
| 463. |
Madison, D. V., and
R. A. Nicoll.
Noradrenaline blocks accommodation of pyramidal cell discharge in the hippocampus.
Nature London
299:
636-638,
1982.
|
| 464. |
Maeda, T.,
C. Pin,
D. Salvert,
L. Leger, and
M. Jouvet.
Les neurones contenant des catecholamines du tegmentum pontique et leurs voies de projection chez le chat.
Brain Res.
57:
119-152,
1973.
|
| 465. |
Maekawa, K., and
D. P. Purpura.
Properties of spontaneous and evoked synaptic activities of thalamic ventrobasal neurons.
J. Neurophysiol.
30:
360-381,
1967.
|
| 466. |
Magni, F., and
W. D. Willis.
Identification of reticular formation neurons by intracellular recording.
Arch. Ital. Biol.
101:
681-702,
1963.
|
| 467. |
Magni, F., and
W. D. Willis.
Cortical control of brain stem reticular neurons.
Arch. Ital. Biol.
102:
418-433,
1964.
|
| 468. |
Magni, F., and
W. D. Willis.
Subcortical and peripheral control of brain stem reticular neurons.
Arch. Ital. Biol.
102:
434-448,
1964.
|
| 469. |
Magoun, H. W., and
R. Rhines.
An inhibitory mechanism in the bulbar reticular formation.
J. Neurophysiol.
9:
165-171,
1946.
|
| 470. |
Malthe‐Sørenssen, D.,
E. Odden, and
I. Walaas.
Selective destruction by kainic acid of neurons innervated by putative glutamergic afferents in septum and nucleus of the diagonal band.
Brain Res.
182:
461-465,
1980.
|
| 471. |
Mancia, M.
Electrophysiological and behavioural changes owing to splitting of the brain stem in cat.
Electroencephalogr. Clin. Neurophysiol.
27:
487-502,
1969.
|
| 472. |
Mancia, M.,
A. Grantyn,
G. Broggi, and
M. Margnelli.
Synaptic linkage between mesencephalic and bulbo‐pontine reticular structures as revealed by intracellular recording.
Brain Res.
33:
491-494,
1971.
|
| 473. |
Mancia, M.,
M. Margnelli,
M. Mariotti,
R. Spreafico, and
G. Broggi.
Brain stem‐thalamus reciprocal influences in the cat.
Brain Res.
69:
297-314,
1974.
|
| 474. |
Mancia, M.,
M. Mariotti,
E. R. Roman, and
M. Schipatti.
Basal forebrain and hypothalamic influences upon brain stem neurons.
Brain Res.
107:
487-497,
1976.
|
| 475. |
Mannen, H.
“Noyau fermé” et “noyau ouvert”. Contribution à l'étude cytoarchitectonique du tronc cérébral envisagée du point de vue du mode d'arborisation dendritique.
Arch. Ital. Biol.
98:
333-350,
1960.
|
| 476. |
Manohar, S.,
H. Noda, and
W. R. Adey.
Behavior of mesencephalic reticular neurons in sleep and wakefulness.
Exp. Neurol.
34:
140-157,
1972.
|
| 477. |
Mantegazzini, P., and
G. Pepeu.
Increase of cortical acetylcholine induced by midbrain hemisection in the cat.
J. Physiol. London
173:
20,
1964.
|
| 478. |
Markowitsch, H. J., and
E. Irle.
Widespread cortical projections of the ventral tegmental area and of other brain stem structures in the cat.
Exp. Brain Res.
41:
233-246,
1981.
|
| 479. |
Marshall, J. F.,
S. Richardson, and
P. Teitelbaum.
Nigrostriatal bundle damage and the lateral hypothalamic syndrome.
J. Comp. Physiol. Psychol.
87:
808-830,
1974.
|
| 480. |
Marshall, J. F.,
B. M. Turner, and
P. Teitelbaum.
Sensory neglect produced by lateral hypothalamic damage.
Science
174:
523-525,
1971.
|
| 481. |
Marshall, K. C., and
H. McLennan.
The synaptic activation of neurones of the feline ventrolateral thalamic nucleus: possible cholinergic mechanisms.
Exp. Brain Res.
15:
472-483,
1972.
|
| 482. |
Marshall, K. C., and
J. S. Murray.
Cholinergic facilitation of thalamic relay transmission in the cat.
Exp. Neurol.
69:
318-333,
1980.
|
| 483. |
Mason, S. T., and
H. C. Fibiger.
Regional topography within noradrenergic locus coeruleus as revealed by retrograde transport of horseradish peroxidase.
J. Comp. Neurol.
187:
703-724,
1979.
|
| 484. |
Mason, S. T., and
H. C. Fibiger.
On the specificity of kainic acid.
Science
204:
1339-1341,
1979.
|
| 485. |
Mason, S. T., and
S. D. Iversen.
Learning in the absence of forebrain noradrenaline.
Nature London
258:
422-424,
1975.
|
| 486. |
Masserano, J. M., and
C. King.
Effects on sleep of acetylcholine perfusion of the locus coeruleus of cats.
Neuropharmacology
21:
1163-1167,
1982.
|
| 487. |
Matsumoto, J., and
M. Jouvet.
Effets de réserpine, dopa et 5‐HTP sur les deux états de sommeil.
C. R. Soc. Biol.
158:
2137-2140,
1964.
|
| 488. |
Matsuzaki, M.
Differential effects of Na‐butyrate and physostigmine on brain stem activities of sleep.
Brain Res.
11:
251-255,
1968.
|
| 489. |
Maunz, R. A.,
N. G. Pitts, and
B. W. Peterson.
Cat spinoreticular neurons: locations, responses and changes in responses during repetitive stimulation.
Brain Res.
148:
365-379,
1978.
|
| 490. |
McBride, R. L., and
J. Sutin.
Projections of the locus coeruleus and adjacent pontine tegmentum in the cat.
J. Comp. Neurol.
165:
265-284,
1976.
|
| 491. |
McCarley, R. W.
Control of sleep‐waking state alteration in Felix domesticus. In:
Aspects of Behavioral Neurobiology,
edited by J. A. Ferrendelli.
Bethesda, MD:
Soc. Neurosci.,
1977,
vol. 3,
p. 90-128.
(Soc. Neurosci. Symp.)
|
| 492. |
McCarley, R. W.,
O. Benoit, and
G. Barrionuevo.
Lateral geniculate nucleus unitary discharge in sleep and waking: state‐ and rate‐specific aspects.
J. Neurophysiol.
50:
798-818,
1983.
|
| 493. |
McCarley, R. W., and
J. A. Hobson.
Single neuron activity in cat gigantocellular tegmental field: selectivity of discharge in desynchronized sleep.
Science
174:
1250-1252,
1971.
|
| 494. |
McCarley, R. W., and
J. A. Hobson.
Simple spike firing patterns of cat cerebellar Purkinje cells in sleep and waking.
Electroencephalogr. Clin. Neurophysiol.
33:
471-483,
1972.
|
| 495. |
McCarley, R. W., and
J. A. Hobson.
Discharge patterns of cat pontine brain stem neurons during desynchronized sleep.
J. Neurophysiol.
38:
751-766,
1975.
|
| 496. |
McCarley, R. W., and
J. A. Hobson.
Neuronal excitability modulation over the sleep cycle: a structural and mathematical model.
Science
189:
58-60,
1975.
|
| 497. |
McCarley, R. W., and
J. A. Hobson.
Mind‐body isomorphism and the study of dreams. In:
Sleep, Dreams and Memory,
edited by W. Fishbein.
New York:
Spectrum,
1981.
|
| 498. |
McCarley, R. W., and
K. Ito.
The pontine reticular formation: an intracellular recording study of inputs from midbrain, bulbar and contralateral pontine reticular formation in the unanesthetized cat.
Soc. Neurosci. Abstr.
6:
203,
1980.
|
| 499. |
McCarley, R. W., and
K. Ito.
Membrane potential depolarization of PRF neurons during behavioral state changes from synchronized to desynchronized sleep in naturally sleeping cats.
Soc. Neurosci. Abstr.
8:
726,
1982.
|
| 500. |
McCarley, R. W.,
J. P. Nelson, and
J. A. Hobson.
Ponto‐geniculo‐occipital (PGO) burst neurons: correlative evidence for neuronal generators of PGO waves.
Science
201:
269-272,
1978.
|
| 501. |
McGinty, D. J., and
R. M. Harper.
Dorsal raphe neurons: depression of firing during sleep in cats.
Brain Res.
101:
569-575,
1976.
|
| 502. |
McGinty, D. J.,
R. M. Harper, and
M. K. Fairbanks.
5‐HT‐containing neurons: unit activity in behaving cats. In:
Serotonin and Behavior,
edited by J. Barchas and
E. Usdin.
New York:
Academic,
1973,
p. 267-269.
|
| 503. |
McGinty, D. J.,
M. S. London,
T. L. Baker,
M. Stevenson,
T. Hoppenbrouwers,
R. M. Harper,
M. B. Sterman, and
J. Hodgman.
Sleep apnea in normal kittens.
Sleep
1:
393-412,
1979.
|
| 504. |
McGinty, D. J., and
J. M. Siegel.
Sleep states. In:
Handbook of Behavioral Neurobiology. Motivation,
edited by E. Satinoff and
P. Teitelbaum.
New York:
Plenum,
1983,
vol. 6,
p. 105-181.
|
| 505. |
McGuiness, C. M., and
G. M. Krauthamer.
The afferent projections to the centrum medianum of the cat as demonstrated by retrograde transport of horseradish peroxidase.
Brain Res.
184:
255-269,
1980.
|
| 506. |
McKenna, T.,
R. W. McCarley,
T. Amatruda,
D. Black, and
J. A. Hobson.
Effects of carbachol at pontine sites yielding long duration desynchronized sleep episodes (Abstract).
Sleep Res.
3:
39,
1974.
|
| 507. |
McMahon, D.
Chemical messengers in development: a hypothesis.
Science
185:
1012-1021,
1974.
|
| 508. |
Meech, R. W.
The sensitivity of Helix aspersa neurones to injected calcium ions.
J. Physiol. London
237:
259-277,
1974.
|
| 509. |
Mehler, W. R.
Further notes on the center median nucleus of Luys. In:
The Thalamus,
edited by D. P. Purpura and
M. D. Yahr.
New York:
Columbia Univ. Press,
1966,
p. 109-122.
|
| 510. |
Menétrey, D.,
A. Chaouch, and
J. M. Besson.
Location and properties of dorsal horn neurons at origin of spinoreticular tract in lumbar enlargement of the rat.
J. Neurophysiol.
44:
862-877,
1980.
|
| 511. |
Mikiten, I. H.,
P. H. Niebyl, and
C. D. Hendley.
EEG desynchronization during behavioral sleep associated with spike discharges from the thalamus of the cat (Abstract).
Federation Proc.
20:
327,
1961.
|
| 512. |
Miller, A. J., and
G. Fry.
Postnatal development of presynaptic terminals in the gigantocellular tegmental field (FTG) of the rat.
Brain Res.
188:
301-317,
1980.
|
| 513. |
Minderhoud, J. M.
An anatomical study of the efferent connections of the thalamic reticular nucleus.
Exp. Brain Res.
12:
435-446,
1971.
|
| 514. |
Mitler, M. M., and
W. C. Dement.
Cataplectic‐like behavior in cats after microinjections of carbachol in pontine reticular formation.
Brain Res.
68:
335-343,
1964.
|
| 515. |
Mitler, M. M., and
W. C. Dement.
Sleep studies on canine narcolepsy: pattern and cycle comparisons between affected and normal dogs.
Electroencephalogr. Clin. Neurophysiol.
43:
691-699,
1977.
|
| 516. |
Mizuno, N.,
C. D. Clemente, and
E. K. Sauerland.
Fiber projections from rostral basal forebrain structures in the cat.
Exp. Neurol.
25:
220-237,
1969.
|
| 517. |
Mizuno, N.,
M. Uemura‐Sumi,
Y. Yasui,
A. Konishi, and
R. Matsushima.
Direct projections from the extrathalamic forebrain structures to the neocortex in the macaque monkey.
Neurosci. Lett.
29:
13-17,
1982.
|
| 518. |
Molliver, M. E.
Role of monoamines in the development of the neocortex.
Neurosci. Res. Program Bull.
20:
492-507,
1982.
|
| 519. |
Molliver, M. E., and
D. A. Kristt.
The fine structural demonstration of monoaminergic synapses in immature rat neocortex.
Neurosci. Lett.
1:
305-310,
1975.
|
| 520. |
Monaco, A. P.,
H. A. Baghdoyan,
J. P. Nelson, and
J. A. Hobson.
Cortical wave amplitude and eye direction movement direction are correlated in REM sleep but not in waking.
Arch. Ital. Biol.
122:
213-223,
1984.
|
| 521. |
Monnier, M.,
A. M. Hatt,
L. B. Cueni, and
G. A. Schoenenberger.
Humoral transmission of sleep. VI. Purification and assessment of a hypnogenic fraction of “sleep Dialysate” (factor delta).
Pfluegers Arch.
331:
257-265,
1972.
|
| 522. |
Montero, V. M., and
G. L. Scott.
Synaptic terminals in the dorsal lateral geniculate nucleus from neurons of the thalamic reticular nucleus: a light and electron microscope autoradiographic study.
Neuroscience
6:
2561-2577,
1981.
|
| 523. |
Moore, R. Y.
The anatomy of central neural mechanisms regulating endocrine rhythms. In:
Endocrine Rhythms,
edited by D. T. Krieger.
New York:
Raven,
1979,
p. 63-87.
|
| 524. |
Moore, R. Y., and
F. E. Bloom.
Central catecholamine neuron systems: anatomy and physiology of the norepinephrine and epinephrine systems.
Annu. Rev. Neurosci.
2:
113-168,
1979.
|
| 525. |
Moore, R. Y.,
A. I. Halaris, and
B. E. Jones.
Serotonin neurons of the midbrain raphe ascending projections.
J. Comp. Neurol.
180:
417-438,
1978.
|
| 526. |
Moore, R. Y., and
D. C. Klein.
Visual pathways and the central neural control of a circadian rhythm in pineal serotonin N‐acetyltransferase activity.
Brain Res.
71:
17-33,
1974.
|
| 527. |
Morales, F., and
M. H. Chase.
Postsynaptic control of lumbar motoneuron excitability during active sleep in the chronic cat.
Brain Res.
225:
279-295,
1981.
|
| 528. |
Morin, D., and
M. Steriade.
Development from primary to augmenting responses in primary somatosensory cortex.
Brain Res.
205:
49-66,
1981.
|
| 529. |
Morison, R. S., and
D. L. Bassett.
Electrical activity of the thalamus and basal ganglia in decorticate cats.
J. Neurophysiol.
8:
309-314,
1945.
|
| 530. |
Morison, R. S., and
E. W. Dempsey.
A study of thalamocortical relations.
Am. J. Physiol.
135:
281-292,
1942.
|
| 531. |
Moroni, F.,
C. Bianchi,
G. Moneti,
S. Tanganelli,
G. Spidelieri,
P. Guandalini, and
L. Beani.
Release of GABA from the guinea‐pig neocortex induced by electrical stimulation of the locus coeruleus or by norepinephrine.
Brain Res.
232:
216-221,
1982.
|
| 532. |
Morrell, J. I.,
L. M. Greenberger, and
D. W. Pfaff.
Hypothalamic, other diencephalic and telencephalic neurons that project to the dorsal midbrain.
J. Comp. Neurol.
201:
589-620,
1981.
|
| 533. |
Morrison, A. R., and
O. Pompeiano.
An analysis of the supraspinal influences acting on motoneurons during sleep in the unrestrained cat. Responses of the alpha motoneurons to direct electrical stimulation during sleep.
Arch. Ital. Biol.
103:
497-516,
1965.
|
| 534. |
Morrison, A. R., and
O. Pompeiano.
Central depolarization of group Ia afferent fibers during desynchronized sleep.
Arch. Ital. Biol.
103:
517-537,
1965.
|
| 535. |
Morrison, A. R., and
O. Pompeiano.
Pyramidal discharge from somatosensory cortex and cortical control of primary afferents during sleep.
Arch. Ital. Biol.
103:
538-568,
1965.
|
| 536. |
Morrison, J. H.,
R. Grzanna,
M. E. Molliver, and
J. T. Coyle.
The distribution and orientation of noradrenergic fibers in neocortex of the rat: an immunofluorescence study.
J. Comp. Neurol.
181:
17-40,
1978.
|
| 537. |
Morrison, J. H.,
M. E. Molliver, and
R. Grzanna.
Noradrenergic innervation of cerebral cortex: widespread effects of local cortical lesions.
Science
205:
313-316,
1979.
|
| 538. |
Moruzzi, G.
Synchronizing influences of the brain stem and the inhibitory mechanisms underlying the production of sleep by sensory stimulation.
Electroencephalogr. Clin. Neurophysiol. Suppl.
13:
231-256,
1960.
|
| 539. |
Moruzzi, G.
The functional significance of sleep with particular regard to the brain mechanisms underlying consciousness. In:
Brain and Conscious Experience,
edited by J. C. Eccles.
New York:
Springer‐Verlag,
1966,
p. 345-388.
|
| 540. |
Moruzzi, G.
Sleep and instinctive behavior.
Arch. Ital. Biol.
107:
175-216,
1969.
|
| 541. |
Moruzzi, G.
The sleep‐waking cycle.
Ergeb. Physiol. Biol. Chem. Exp. Pharmakol.
64:
1-165,
1972.
|
| 542. |
Moruzzi, G., and
H. W. Magoun.
Brainstem reticular formation and activation of the EEG.
Electroencephalogr. Clin. Neurophysiol.
1:
455-473,
1949.
|
| 543. |
Motter, B. C., and
V. B. Mountcastle.
The functional properties of the light‐sensitive neurons of the posterior parietal cortex studied in waking monkeys: foveal sparing and opponent vector organization.
J. Neurosci.
1:
3-26,
1981.
|
| 544. |
Mountcastle, V. B.,
R. A. Andersen, and
B. C. Motter.
The influence of attentive fixation upon the excitability of the light‐sensitive neurons of the posterior parietal cortex.
J. Neurosci.
1:
1218-1235,
1981.
|
| 545. |
Mountcastle, V. B.,
J. C. Lynch,
A. Georgopoulos,
H. Sakata, and
C. Acuna.
Posterior parietal association cortex of the monkey: command functions for operations within extrapersonal space.
J. Neurophysiol.
38:
871-908,
1975.
|
| 546. |
Mouret, J., and
J. Coindet.
Polygraphic evidence against a critical role of the raphe nuclei in sleep in the rat.
Brain Res.
186:
273-287,
1980.
|
| 547. |
Mouret, J.,
J. Coindet,
G. Debilly, and
G. Chouvet.
Suprachiasmatic nuclei lesions in the rat: alterations in sleep circadian rhythms.
Electroencephalogr. Clin. Neurophysiol.
45:
402-408,
1978.
|
| 548. |
Mukhametov, L. M.,
G. Rizzolatti, and
A. Seitun.
An analysis of the spontaneous activity of lateral geniculate neurons and of optic tract fibers in free moving cats.
Arch. Ital. Biol.
108:
325-347,
1970.
|
| 549. |
Mukhametov, L. M.,
G. Rizzolatti, and
V. Tradardi.
Spontaneous activity of nucleus reticularis thalami in freely moving cats.
J. Physiol. London
210:
651-667,
1970.
|
| 550. |
Nakahama, H.,
K. Shima,
M. Yamamoto, and
K. Aya.
Regularity of the spontaneous discharge of neurons in the nucleus raphe dorsalis of the cat.
Neurosci. Lett.
23:
161-165,
1981.
|
| 551. |
Nakai, Y., and
S. Takaori.
Influence of norepinephrine‐containing neurons derived from the locus coeruleus on lateral geniculate neuronal activities of cats.
Brain Res.
71:
47-60,
1974.
|
| 552. |
Nakamura, R. H.,
C. Kennedy,
J. C. Fillin,
S. Suda,
M. Ito,
F. Storch,
W. Mendelson,
M. Mishkin, and
L. Sokoloff.
Hypnogenic‐center theory of sleep: no support from 2‐deoxyglucose studies in monkeys.
Soc. Neurosci. Abstr.
7:
234,
1981.
|
| 553. |
Nakamura, S., and
K. Iwama.
Antidromic activation of the rat locus coeruleus neurons from hippocampus, cerebral and cerebellar cortices.
Brain Res.
99:
372-376,
1975.
|
| 554. |
Nakamura, Y.,
L. Goldberg,
S. Chandler, and
M. Chase.
Intracellular analysis of trigeminal motoneuron activity during sleep in the cat.
Science
199:
204-207,
1978.
|
| 555. |
Nakamura, Y., and
J. Schlag.
Cortically induced rhythmic activities in the thalamic ventrolateral nucleus of the cat.
Exp. Neurol.
22:
209-221,
1968.
|
| 556. |
Nakano, K.,
T. Takimoto,
T. Kayahara,
Y. Takeuchi, and
Y. Kobaysahi.
Distribution of cerebellothalamic neurons projecting to the ventral nuclei of the thalamus: an HRP study in the cat.
J. Comp. Neurol.
194:
427-439,
1980.
|
| 557. |
Nakazawa, Y.,
H. Tachibana,
M. Kotorii, and
M. Ogata.
Effects of L‐dopa on natural night sleep and on rebound of REM sleep.
Folia Psychiatr. Neurol. Jpn.
27:
223-230,
1973.
|
| 558. |
Nauta, W. J. H.
Hippocampal projections and related neural pathways to the midbrain in the cat.
Brain
81:
319-340,
1958.
|
| 559. |
Nauta, W. J. H.
Projections of the pallidal complex: an autoradiographic study in the cat.
Neuroscience
4:
1853-1873,
1979.
|
| 560. |
Nauta, W. J. H., and
M. Cole.
Efferent projections of the subthalamic nucleus: an autoradiographic study in monkey and cat.
J. Comp. Neurol.
180:
1-16,
1978.
|
| 561. |
Nauta, W. J. H., and
H. G. J. M. Kuypers.
Some ascending pathways in the brain stem reticular formation. In:
Reticular Formation of the Brain,
edited by H. H. Jasper,
L. D. Proctor,
R. S. Knighton,
W. C. Noshay, and
R. T. Costello.
Boston, MA:
Little, Brown,
1958,
p. 3-30.
|
| 562. |
Nauta, W. J. H., and
W. R. Mehler.
Projections of the lentiform nucleus in the monkey.
Brain Res.
1:
3-42,
1966.
|
| 563. |
Nauta, H. J. W.,
M. B. Pritz, and
R. J. Lassek.
Afferents to the rat caudatoputamen studied with horseradish peroxidase: an evaluation of a retrograde neuroanatomical research method.
Brain Res.
67:
219-238,
1974.
|
| 564. |
Nelson, J. P.,
R. W. McCarley, and
J. A. Hobson.
REM sleep burst neurons, PGO waves, and eye movement information.
J. Neurophysiol.
50:
784-797,
1983.
|
| 565. |
Netick, A.,
J. Orem, and
W. Dement.
Neuronal activity specific to REM sleep and its relationship to breathing.
Brain Res.
120:
197-207,
1977.
|
| 566. |
Nicoll, R. A., and
B. E. Alger.
Synaptic excitation may activate a calcium‐dependent potassium conductance in hippocampal pyramidal cells.
Science
212:
957-959,
1981.
|
| 567. |
Nieouellon, A.,
A. Cheramy, and
J. Glowinski.
Release of dopamine in vivo from cat substantia nigra.
Nature London
266:
375-377,
1977.
|
| 568. |
Niimi, K.,
S. Kishi,
M. Miki, and
S. Fujita.
An experimental study of the course and termination of the projection fibers from cortical areas 4 and 6 in the cat.
Folia Psychiatr. Neurol. Jpn.
17:
167-216,
1963.
|
| 569. |
Niimi, K., and
E. Kuwahara.
The dorsal thalamus of the cat and comparison with monkey and man.
J. Hirnforsch.
14:
303-325,
1973.
|
| 570. |
Niimi, K.,
M. Niimi, and
Y. Okada.
Thalamic afferents to the limbic cortex in the cat studied with the method of retrograde axonal transport of horseradish peroxidase.
Brain Res.
145:
225-238,
1978.
|
| 571. |
Oakson, G., and
M. Steriade.
Slow rhythmic rate fluctuations of cat midbrain reticular neurons in synchronized sleep and waking.
Brain Res.
247:
277-288,
1982.
|
| 572. |
Oderfeld‐Nowak, B.,
A. Potempska, and
J. Oderfeld.
Analysis of the time course of changes in hippocampal acetylcholinesterase and choline acetyltransferase activities after various septal lesions in the rat: return of enzymic activity after extensive medioventral lesions.
Neuroscience
2:
641-648,
1977.
|
| 573. |
Oertel, W. H.,
A. M. Graybiel,
E. Mugnaini,
R. P. Elde,
D. E. Schmechel, and
I. J. Kopin.
Coexistence of glutamic acid decarboxylase and somatostatinlike immunoreactivity in neurons of the feline nucleus reticularis thalami.
J. Neurosci.
3:
1322-1332,
1983.
|
| 574. |
Ohara, P. T., and
A. R. Liebermann.
Thalamic reticular nucleus: anatomical evidence that cortico‐reticular axons establish monosynaptic contact with reticulo‐geniculate projection cells.
Brain Res.
207:
153-156,
1981.
|
| 575. |
Ohara, P. T.,
A. J. Sefton, and
A. R. Liebermann.
Mode of termination of afferents from the thalamic reticular nucleus in the dorsal lateral geniculate nucleus of the rat.
Brain Res.
197:
503-506,
1980.
|
| 576. |
Olivier, A.,
A. Parent, and
L. J. Poirier.
Identification of the thalamic nuclei on the basis of their cholinesterase content in the monkey.
J. Anat.
106:
37-50,
1970.
|
| 577. |
Olschowska, J. A.,
R. Grzanna, and
M. E. Molliver.
The distribution and incidence of synaptic contacts of noradrenergic varicosities in the rat neocortex: an immunocytochemical study.
Soc. Neurosci. Abstr.
6:
352,
1980.
|
| 578. |
Olson, L., and
K. Fuxe.
On the projections from the locus coeruleus noradrenaline neurons: the cerebellar innervation.
Brain Res.
28:
165-171,
1971.
|
| 579. |
Olszewski, J., and
D. Baxter.
The Cytocarchitecture of the Human Brain Stem.
New York:
Karger,
1954.
|
| 580. |
Oshtory, M. A., and
N. Vijayan.
Clorazepam treatment of insomnia due to sleep myoclonus.
Arch. Neurol.
37:
119-120.
1980.
|
| 581. |
Oswald, I.
Human brain protein drugs and dreams.
Nature London
223:
893-897,
1969.
|
| 582. |
Oswald, I.,
K. Adam,
S. Allen,
R. Burack,
M. Spence, and
V. Thacore.
Alpha adrenergic blocker, thymoxamine and mesoridaxine both increase human REM sleep duration (Abstract).
Sleep Res.
3:
62,
1974.
|
| 583. |
Oswald, I.,
V. R. Thacore,
K. Adam,
V. Brezinova, and
R. Burack.
α‐Adrenergic blockade increases human REM sleep.
Br. J. Clin. Pharmacol.
2:
107-110,
1975.
|
| 584. |
Ottersen, O. P., and
Y. Ben‐Ari.
Demonstration of a heavy projection of midline thalamic neurons upon the lateral nucleus of the amygdala of the rat.
Neurosci. Lett.
9:
147-152,
1978.
|
| 585. |
Pappenheimer, J. R.,
G. Koski,
V. Fencl,
M. L. Karnovsky, and
J. Krueger.
Extraction of sleep‐promoting factor S from cerebrospinal fluid and from brains of sleep‐deprived animals.
J. Neurophysiol.
38:
1299-1311,
1975.
|
| 586. |
Parent, A.
Comparative anatomy of monoaminergic systems. In:
Handbook of Chemical Neuroanatomy,
edited by A. Björklund,
T. Hökfelt, and
M. Kuhar.
Amsterdam:
Elsevier,
1984,
vol. 2,
p. 409-439.
|
| 587. |
Parent, A.,
L. Descarries, and
A. Beaudet.
Organization of ascending serotonin systems in the adult rat brain. A radioautoradiographic study after intraventricular administration of [3H]5‐hydroxytryptamine.
Neuroscience
6:
115-138,
1981.
|
| 588. |
Parent, A., and
M. Steriade.
Afferents from the periaqueductal gray, medial hypothalamus and medial thalamus to the midbrain reticular core.
Brain Res. Bull.
7:
411-418,
1981.
|
| 589. |
Parent, A., and
M. Steriade.
Midbrain tegmental projections of nucleus reticularis thalami of cat and monkey: a retrograde transport and antidromic identification study.
J. Comp. Neurol.
229:
548-558,
1984.
|
| 590. |
Partlow, G. D.,
M. Colonnier, and
J. Szabo.
Thalamic projections of the superior colliculus in the rhesus monkey, Macaca mulatto. A light and electron microscopic study.
J. Comp. Neurol.
171:
285-318,
1977.
|
| 591. |
Pasik, P.,
T. Pasik, and
J. Hamori.
Synapses between interneurons in the lateral geniculate nucleus of monkeys.
Exp. Brain Res.
25:
1-13,
1976.
|
| 592. |
Pasik, P.,
T. Pasik,
J. Hamori, and
J. Szentagothai.
Golgi type II interneurons in the neuronal circuit of the monkey lateral geniculate nucleus.
Exp. Brain Res.
17:
18-34,
1973.
|
| 593. |
Passouant, P.
Problèmes physiopathologiques de la narcolepsie et periodicite du “sommeil rapide” au cours du nycthemere. In:
The Abnormalities of Sleep in Man,
edited by H. Gastaut,
E. Lugaresi,
G. Berti Ceroni, and
G. Coccagna.
Bologna, Italy:
Gaggi,
1968,
p. 177-189.
|
| 594. |
Pavlov, I. P.
Innere Hemmung der Bedingten Reflexe und der Schlaf—ein und derselbe Prozess.
Skand. Arch. Physiol.
44:
42-58,
1923.
|
| 595. |
Pavlov, I. P.
Conditioned Reflexes. An Investigation of the Physiological Activity of the Cerebral Cortex,
translated by G. V. Anrep.
New York:
Dover,
1960.
|
| 596. |
Payne, B. R., and
N. Berman.
Contralateral corticofugal projections to cat visual thalamus.
Exp. Brain Res.
53:
462-466,
1984.
|
| 597. |
Pearce, G. W.
Some cortical projections to the midbrain reticular formation. In:
Structure and Function of the Cerebral Cortex,
edited by D. B. Tower and
J. P. Schadé
Amsterdam:
Elsevier,
1960,
p. 131-137.
|
| 598. |
Pepeu, G., and
P. Mantegazzani.
Midbrain hemisection: effect on cortical acetylcholine in the cat.
Science
145:
1069-1070,
1964.
|
| 599. |
Peterson, B. W.
Identification of reticulospinal projections that may participate in gaze control. In:
Control of Gaze by Brain Stem Neurons,
edited by R. Baker and
A. Berthoz.
Amsterdam:
Elsevier,
1977,
p, 143-152.
|
| 600. |
Peterson, B. W.,
M. E. Anderson, and
M. Filion.
Responses of pontomedullary reticular neurons to cortical, tectal and cutaneous stimuli.
Exp. Brain Res.
21:
19-44,
1974.
|
| 601. |
Peterson, B. W.,
J. I. Franck,
N. G. Pitts, and
N. G. Daunton.
Changes in responses of medial pontomedullary reticular neurons during repetitive cutaneous, vestibular, cortical and tectal stimulation.
J. Neurophysiol.
39:
564-581,
1976.
|
| 602. |
Peterson, B. W.,
R. A. Maunz,
N. G. Pitts, and
R. G. Mackel.
Patterns of projection and branching of reticulospinal neurons.
Exp. Brain Res.
23:
333-351,
1975.
|
| 603. |
Peterson, B. W.,
N. G. Pitts,
K. Fukushima, and
R. Mackel.
Reticulospinal excitation and inhibition of neck motoneurons.
Exp. Brain Res.
32:
471-489,
1978.
|
| 604. |
Petras, J. M.
Some efferent connections of the motor and somatosensory cortex of simian primates and felid, canid and procyonid carnivores.
Ann. NY Acad. Sci.
167:
469-505,
1969.
|
| 605. |
Petsche, H.,
G. Gogolak, and
P. A. Van Zwieten.
Rhythmicity of septal cell discharges at various levels of reticular excitation.
Electroencephalogr. Clin. Neurophysiol.
19:
25-33,
1965.
|
| 606. |
Phillipson, E. A.
Control of breathing during sleep.
Am. Rev. Respir. Dis.
118:
909-939,
1978.
|
| 607. |
Phillipson, E. A., and
C. E. Sullivan.
Respiratory control mechanisms during NREM and REM sleep. In:
Sleep Apnea Syndromes,
edited by C. Guilleminault and
W. C. Dement.
New York:
Liss,
1978,
p. 47-74.
|
| 608. |
Phillipson, E. A.,
C. E. Sullivan,
D. J. C. Read,
E. Murphy, and
L. F. Kozar.
Ventilatory and waking responses to hypoxia in sleeping dogs.
J. Appl. Physiol.: Respirat. Environ. Exercise Physiol.
44:
512-520,
1978.
|
| 609. |
Phillipson, O. T.
Afferent projections to the ventral tegmental area of Tsai and interfascicular nucleus: a horseradish peroxidase study in the rat.
J. Comp. Neurol.
187:
117-144,
1979.
|
| 610. |
Phillis, J. W.
Acetylcholine release from the cerebral cortex: its role in cortical arousal.
Brain Res.
7:
378-389,
1968.
|
| 611. |
Phillis, J. W., and
G. C. Chong.
Acetylcholine release from the cerebral and cerebellar cortices: its role in cortical arousal.
Nature London
207:
1253-1255,
1965.
|
| 612. |
Phillis, J. W., and
G. K. Kostopoulos.
Activation of a noradrenergic pathway from the brain stem to rat neocortex.
Gen. Pharmacol.
8:
207-211,
1977.
|
| 613. |
Phillis, J. W., and
D. H. York.
Pharmacological studies on a cholinergic inhibition in the cerebral cortex.
Brain Res.
10:
297-306,
1968.
|
| 614. |
Pickel, V. M.,
M. Segal, and
F. E. Bloom.
A radioautographic study of the efferent pathways of the nucleus locus coeruleus.
J. Comp. Neurol.
155:
15-42,
1974.
|
| 615. |
Pivik, R. T.,
R. W. McCarley, and
J. A. Hobson.
Eye movement‐associated discharge in brain stem neurons during desynchronized sleep.
Brain Res.
121:
59-76,
1977.
|
| 616. |
Poitras, D., and
A. Parent.
Atlas of the distribution of monoamine‐containing nerve cell bodies in the brain stem of the cat.
J. Comp. Neurol.
179:
699-718,
1978.
|
| 617. |
Pompeiano, O.
The neurophysiological mechanisms of the postural and motor events during desynchronized sleep.
Proc. Assoc. Res. Nerv. Ment. Dis.
45:
351-423,
1967.
|
| 618. |
Pompeiano, O.
Reticular formation. In:
Handbook of Sensory Physiology. Somatosensory System,
edited by A. Iggo.
Berlin:
Springer‐Verlag,
1973,
vol. II,
p. 381-488.
|
| 619. |
Pompeiano, O.
Cholinergic activation of reticular and vestibular mechanisms controlling posture and eye movements. In:
The Reticular Formation Revisited,
edited by J. A. Hobson and
M. A. B. Brazier.
New York:
Raven,
1979,
p. 473-572.
|
| 620. |
Pompeiano, O., and
C. D. Barnes.
Response of brain stem reticular neurons to muscle vibration in the decerebrate cat.
J. Neurophysiol.
34:
709-724,
1971.
|
| 621. |
Pompeiano, O., and
K. Hoshino.
Central control of posture: reciprocal discharge by two pontine neuronal groups leading to suppression of decerebrate rigidity.
Brain Res.
116:
131-138,
1976.
|
| 622. |
Pompeiano, O., and
A. R. Morrison.
Vestibular influences during sleep. I. Abolition of the rapid eye movements of desynchronized sleep following vestibular lesions.
Arch. Ital. Biol.
103:
569-595,
1965.
|
| 623. |
Pompeiano, O., and
M. Valentinuzzi.
A mathematical model for the mechanism of rapid eye movements induced by an anticholinesterase in the decerebrate cat.
Arch. Ital. Biol.
114:
103-154,
1976.
|
| 624. |
Porrino, L. J., and
P. S. Goldman‐Rakic.
Brain stem innervation of prefrontal and anterior cingulate cortex in the rhesus monkey revealed by retrograde transport of HRP.
J. Comp. Neurol.
205:
63-76,
1982.
|
| 625. |
Powell, T. P. S., and
W. M. Cowan.
The interpretation of the degenerative changes in the intralaminar nuclei of the thalamus.
J. Neurol. Neurosurg. Psychiatry
30:
140-153,
1967.
|
| 626. |
Powles, A. C. P.,
J. R. Sutton,
G. W. Gray,
A. L. Mansell,
M. McFadden, and
C. S. Houston.
Sleep hypoxemia at altitude: its relationship to acute mountain sickness and ventilatory responsiveness to hypoxia and hypercapnia. In:
Environmental Stress,
edited by L. J. Folinsbee.
New York:
Academic,
1978,
p. 373-381.
(Proc. Int. Symp. Environ. Stress.)
|
| 627. |
Puizillout, J. J.,
G. Gaudin‐Chazal,
A. Daszuta,
N. Seyfritz, and
J. P. Ternaux.
Release of endogenous serotonin from “encephale isolé” cats. II. Correlations with raphe neuronal activity and sleep and wakefulness.
J. Physiol. Paris
75:
531-537,
1979.
|
| 628. |
Puizillout, J. J., and
J. P. Ternaux.
Endormement vago‐aortique après section sagittale médiane du tronc cérébral et après administration de p‐chlorophenylanaline ou destruction de noyaux du raphé.
Brain Res.
70:
19-42,
1974.
|
| 629. |
Purpura, D. P.
Interneuronal mechanisms in thalamically induced synchronizing and desynchronizing activities. In:
The Interneuron,
edited by M. A. B. Brazier.
Berkeley:
Univ. of California Press,
1969,
p. 467-496.
|
| 630. |
Purpura, D. P.
Operations and processes in thalamic and synaptically related neural subsystems. In:
The Neurosciences: Second Study Program,
edited by F. O. Schmitt.
New York:
Rockefeller Univ. Press,
1970.
p. 458-470.
|
| 631. |
Purpura, D. P.
Intracellular studies of synaptic organizations in the mammalian brain. In:
Structure and Function of Synapses,
edited by G. D. Pappas and
D. P. Purpura.
New York:
Raven,
1972,
p. 257-302.
|
| 632. |
Purpura, D. P.,
J. G. McMurtry, and
K. Maekawa.
Synaptic events in ventro‐lateral thalamic neurons during suppression of recruiting responses by brain stem reticular stimulation.
Brain Res.
1:
63-76,
1966.
|
| 633. |
Purpura, D. P., and
R. J. Shofer.
Intracellular recording from thalamic neurons during reticulocortical activation.
J. Neurophysiol.
26:
494-505,
1963.
|
| 634. |
Purpura, D. P.,
R. J. Shofer, and
F. S. Musgrave.
Cortical intracellular potentials during augmenting and recruiting responses. II. Patterns of synaptic activities in pyramidal and nonpyramidal tract neurons.
J. Neurophysiol.
27:
133-151,
1964.
|
| 635. |
Rackowski, D., and
I. T. Diamond.
Cortical connections of the pulvinar nucleus in Galago.
J. Comp. Neurol.
193:
1-40,
1980.
|
| 636. |
Ramon‐Moliner, E.
La différenciation morphologique des neurones.
Arch. Ital. Biol.
105:
149-188,
1967.
|
| 637. |
Ramon‐Moliner, E., and
W. J. H. Nauta.
The isodendritic core of the brain stem.
J. Comp. Neurol.
126:
311-336,
1966.
|
| 638. |
Ramón y Cajal, S.
Histologie du système nerveux de l'homme et des vertébrés,
translated by S. Azoulay.
Paris:
Maloine,
1909‐1911,
vols. I and II.
|
| 639. |
Ranck, J. B.
Studies on single neurons in dorsal hippocampal formation and septum in unrestrained rats. I. Behavioral correlates and firing repertoires.
Exp. Neurol.
41:
461-531,
1973.
|
| 640. |
Rasminski, M.,
A. J. Mauro, and
D. Albe‐Fessard.
Projection of medial thalamic nuclei to putamen and cerebral frontal cortex in the cat.
Brain Res.
61:
69-77,
1973.
|
| 641. |
Reader, T. A.,
A. Ferron,
L. Descarries, and
H. H. Jasper.
Modulatory role for biogenic amines in the cerebral cortex. Microiontophoretic studies.
Brain Res.
160:
217-229,
1979.
|
| 642. |
Rechtschaffen, A.,
R. A. Lovell,
D. Freedman,
W. E. Whitehead, and
M. Aldrich.
The effect of parachlorophenylalamine on sleep in the rat: some implications for the serotonin sleep hypothesis. In:
Serotonin and Behavior,
edited by J. Barchas and
E. Usdin.
New York:
Academic,
1973,
p. 401-418.
|
| 643. |
Reed, D. J., and
R. H. Kellogg.
Changes in respiratory response to CO2 during natural sleep at sea level and at altitude.
J. Appl. Physiol.
13:
325-330,
1958.
|
| 644. |
Reed, D. Jr. and
R. H. Kellogg.
Effect of sleep on hypoxic stimulation of breathing at sea level and altitude.
J. Appl. Physiol.
15:
1130-1134,
1960.
|
| 645. |
Reed, D. J., and
R. H. Kellogg.
Effect of sleep on CO2 stimulation of breathing in acute and chronic hypoxia.
J. Appl. Physiol.
15:
1135-1138,
1960.
|
| 646. |
Reeves, A. G., and
W. D. Hagamen.
Behavioral and EEG asymmetry following unilateral lesions of the forebrain and midbrain in cats.
Electroencephalogr. Clin. Neurophysiol.
30:
83-86,
1971.
|
| 647. |
Reite, M.,
D. Jackson, and
R. L. Cahoon.
Sleep physiology at high altitude.
Electroencephalogr. Clin. Neurophysiol.
38:
463-471,
1975.
|
| 648. |
Reite, M.,
G. V. Pegram,
L. M. Stephens,
E. C. Bixler, and
O. L. Lewis.
The effect of reserpine and monoamine oxidase inhibitors on paradoxical sleep in the monkey.
Psychopharrnacologia
14:
12-17,
1969.
|
| 649. |
Reppert, S. M.,
M. J. Perlow,
L. G. Ungerleider,
M. Mishkin,
L. Tamarkin,
D. G. Orloff,
H. J. Hoffman, and
D. C. Klein.
Effects of damage to the suprachiasmatic area of the anterior hypothalamus on the daily melatonin and Cortisol rhythms in the rhesus monkey.
J. Neurosci.
1:
1414-1425,
1981.
|
| 650. |
Ribak, C. E.
Aspinous and sparsely‐spinous stellate neurons in the visual cortex of rats contain glutamic acid decarboxylase.
J. Neurocytol.
7:
461-478,
1978.
|
| 651. |
Richards, J. G.,
H. P. Lorez, and
J. P. Tranzer.
Indoleal‐kylamine nerve terminals in cerebral ventricles: identification by electron microscopy and fluorescence histochemistry.
Brain Res.
57:
277-288,
1973.
|
| 652. |
Richmond, B. J., and
R. H. Wurtz.
Constriction of visual receptive fields of inferior temporal cortex neurons during visual fixation.
Invest. Ophthal. Visual Sci.
20:
148,
1981.
|
| 653. |
Richter, C. P.
Biological Clocks in Medicine and Psychiatry.
Springfield, IL:
Thomas,
1965.
|
| 654. |
Rinvik, E.
The corticothalamic projection from the pericruciate and coronal gyri in the cat: an experimental study with silver‐impregnation methods.
Brain Res.
10:
79-119,
1968.
|
| 655. |
Rinvik, E.
Organization of thalamic connections from motor and somatosensory cortical areas in the cat. In:
Corticothalamic Projections and Sensorimotor Activities,
edited by T. Frigyesi,
E. Rinvik, and
M. D. Yahr.
New York:
Raven,
1972,
p. 57-90.
|
| 656. |
Rinvik, E.
Demonstration of nigrothalamic connections in the cat by retrograde axonal transport of horseradish peroxidase.
Brain Res.
90:
313-318,
1975.
|
| 657. |
Ritvo, E. R.,
E. M. Ornitz,
S. La Franchi, and
R. D. Walter.
Effects of imipramine on the sleep‐dream cycle: an EEG study in boys.
Electroencephalogr. Clin. Neurophysiol.
22:
465-468,
1967.
|
| 658. |
Rivner, M., and
J. Sutin.
Locus coeruleus modulation of the motor thalamus: inhibition in nuclei ventralis lateralis and ventralis anterior.
Exp. Neurol.
73:
651-673,
1981.
|
| 659. |
Robertson, R. T., and
T. J. Cunningham.
Organization of corticothalamic projections from parietal cortex in cat.
J. Comp. Neurol.
199:
569-585,
1981.
|
| 660. |
Robertson, R. T., and
A. R. Feiner.
Diencephalic projections from the pontine reticular formation: autoradiographic studies in the cat.
Brain Res.
239:
3-16,
1982.
|
| 661. |
Robertson, R. T., and
S. S. Kaitz.
Thalamic connections with limbic cortex. I. Thalamocortical projections.
J. Comp. Neurol.
195:
501-525,
1981.
|
| 662. |
Robertson, R. T.,
G. S. Lynch, and
R. F. Thompson.
Diencephalic distributions of ascending reticular systems.
Brain Res.
55:
309-322,
1973.
|
| 663. |
Robinson, D. A.
Models of oculomotor neural organization in the control of eye movements. In:
Basic Mechanisms of Ocular Motility and Their Clinical Implications,
edited by G. Lennerstrand and
P. Bach‐y‐Rita.
Oxford, UK:
Pergamon,
1975,
p. 519-538.
(Wenner‐Gren Ctr. Int. Symp. 1974.)
|
| 664. |
Robinson, T. E.,
C. H. Vanderwolf, and
B. A. Pappas.
Are the dorsal noradrenergic bundle projections from the locus coeruleus important for neocortical or hippocampal activation?
Brain Res.
138:
75-98,
1977.
|
| 665. |
Roffwarg, H. P.,
J. N. Muzio, and
W. C. Dement.
Ontogenetic development of the human sleep‐dream cycle.
Science
152:
604-619,
1966.
|
| 666. |
Room, P.,
F. Postema, and
J. Korf.
Divergent axon collaterals of rat locus coeruleus neurons: demonstration by a fluorescent double labeling technique.
Brain Res.
221:
219-230,
1981.
|
| 667. |
Ropert, N., and
M. Steriade.
Input‐output organization of the midbrain reticular core.
J. Neurophysiol.
46:
17-31,
1981.
|
| 668. |
Rose, J. E., and
V. B. Mountcastle.
The thalamic tactile region in rabbit and cat.
J. Comp. Neurol.
97:
441-489,
1952.
|
| 669. |
Rossi, G. F., and
A. Brodal.
Corticofugal fibres to the brain stem reticular formation: an experimental study in the cat.
J. Anal
90:
42-62,
1956.
|
| 670. |
Rossor, M. N.,
C. Svendsen,
S. P. Hunt,
C. Q. Mountjoy,
M. Roth, and
L. L. Iversen.
The substantia innominata in Alzheimer's disease: an histochemical and biochemical study of cholinergic marker enzymes.
Neurosci. Lett.
28:
217-222,
1982.
|
| 671. |
Roth, S. R.,
M. B. Sterman, and
C. D. Clemente.
Comparison of EEG correlates of reinforcement, internal inhibition and sleep.
Electroencephalogr. Clin. Neurophysiol.
23:
509-520,
1967.
|
| 672. |
Rougeul‐Buser, A.,
J. J. Bouyer, and
P. Buser.
From attentiveness to sleep. A topographical analysis of localized “synchronized” activities on the cortex of normal cat and monkey.
Acta Neurobiol. Exp.
35:
805-819,
1975.
|
| 673. |
Rougeul‐Buser, A.,
J. J. Bouyer,
M. F. Montaron, and
P. Buser.
Patterns of activities in the ventrobasal thalamus and somatic cortex SI during behavioral waking immobility in the cat.
Exp. Brain Res. Suppl.
7:
69-87,
1983.
|
| 674. |
Roy, J. P.,
M. Clercq,
M. Steriade, and
M. Deschěnes.
Electrophysiology of neurons of lateral thalamic nuclei in cat: mechanisms of long‐lasting hyperpolarizations.
J. Neurophysiol.
51:
1220-1235,
1984.
|
| 675. |
Roy, J. P.,
J. Hada, and
M. Steriade.
The fastigio‐ventro‐medial thalamic‐cortical projection.
Soc. Neurosci. Abstr.
7:
414,
1981.
|
| 676. |
Royce, G. J.
Single thalamic neurons which project to both the rostral cortex and caudate nucleus studied with the fluorescent double labeling method.
Exp. Neurol.
79:
773-784,
1983.
|
| 677. |
Ruch‐Monachon, M. A.,
M. Jalfre, and
W. E. Haefely.
Drugs and PGO waves in the lateral geniculate body of the curarized cat. I. PGO wave activity induced by Ro 43284 and by p‐chlorophenylalanine (PCPA) as a basis for neuropharmacological studies.
Arch. Int. Pharmacodyn. Ther.
219:
251-268,
1976.
|
| 678. |
Ruch‐Monachon, M. A.,
M. Jalfre, and
W. E. Haefely.
Drugs and PGO waves in the lateral geniculate body of the curarized cat. II. PGO wave activity and brain 5‐hydroxytryptamine.
Arch. Int. Pharmacodyn. Ther.
219:
269-286,
1976.
|
| 679. |
Ruch‐Monachon, M. A.,
M. Jalfre, and
W. E. Haefely.
Drugs and PGO waves in the lateral geniculate body of the curarized cat. III. PGO wave activity and brain catecholamines.
Arch. Int. Pharmacodyn. Ther.
219:
287-307,
1976.
|
| 680. |
Ruch‐Monachon, M. A.,
M. Jalfre, and
W. E. Haefely.
Drugs and PGO waves in the lateral geniculate body of the curarized cat. IV. The effects of acetylcholine, GABA and benzodiapines on PGO wave activity.
Arch. Int. Pharmacodyn. Ther.
219:
308-325,
1976.
|
| 681. |
Ruch‐Monachon, M. A.,
M. Jalfre, and
W. E. Haefely.
Drugs and PGO waves in the lateral geniculate body of the curarized cat. V. Miscellaneous compounds. Synopsis of the role of central neurotransmitters on PGO wave activity.
Arch. Int. Pharmacodyn. Ther.
219:
326-346,
1976.
|
| 682. |
Rusak, B.
The role of the suprachiasmatic nuclei in the generation of circadian rhythms in the golden hamster, Mesocricetus auratus.
J. Comp. Physiol.
118:
145-164,
1977.
|
| 683. |
Rusak, B., and
G. Groos.
Suprachiasmatic stimulation phase shifts rodent circadian rhythms.
Science
215:
1407-1409,
1982.
|
| 684. |
Rusak, B., and
I. Zucker.
Neural regulation of circadian rhythms.
Physiol. Rev.
59:
449-526,
1979.
|
| 685. |
Saito, H.,
K. Sakai, and
M. Jouvet.
Discharge patterns of the nucleus parabrachialis lateralis neurons of the cat during sleep and waking.
Brain Res.
134:
59-72,
1977.
|
| 686. |
Sakai, F.,
J. S. Meyer,
I. Karacan,
F. Yamaguchi, and
M. Yamamoto.
Narcolepsy: regional cerebral blood flow during sleep and wakefulness.
Neurology
29:
61-67,
1979.
|
| 687. |
Sakai, K.
Some anatomical and physiological properties of pontomesencephalic tegmental neurons with special reference to the PGO waves and postural atonia during paradoxical sleep in the cat. In:
The Reticular Formation Revisited,
edited by J. A. Hobson and
M. A. B. Brazier.
New York:
Raven,
1979,
p. 427-447.
|
| 688. |
Sakai, K., and
M. Jouvet.
Brain stem PGO‐on cells projecting directly to the cat dorsal lateral geniculate nucleus.
Brain Res.
194:
500-505,
1980.
|
| 689. |
Sakai, K.,
D. Salvert,
M. Touret, and
M. Jouvet.
Afferent connections of the nucleus raphe dorsalis in the cat as visualized by the horseradish peroxidase technique.
Brain Res.
137:
11-35,
1977.
|
| 690. |
Sakai, K.,
J. P. Sastre,
N. Kanamori, and
M. Jouvet.
State‐specific neurons in the ponto‐medullary reticular formation with special reference to the postural atonia during paradoxical sleep in the cat. In:
Brain Mechanisms of Perceptual Awareness and Purposeful Behavior,
edited by O. Pompeiano and
C. Ajmone‐Marsan.
New York:
Raven,
1981,
p. 405-429.
|
| 691. |
Sakai, K.,
J. P. Sastre,
D. Salvert,
M. Touret,
M. Tohyama, and
M. Jouvet.
Tegmentoreticular projections with special reference to the muscular atonia during paradoxical sleep in the cat: an HRP study.
Brain Res.
176:
233-254,
1979.
|
| 692. |
Sakai, K.,
M. Touret,
D. Salvert,
L. Leger, and
M. Jouvet.
Afferent projections to the cat locus coeruleus as visualized by the horseradish peroxidase technique.
Brain Res.
119:
21-41,
1977.
|
| 693. |
Sakakura, H.
Spontaneous and evoked unitary activities of cat lateral geniculate neurons in sleep and wakefulness.
Jpn. J. Physiol.
18:
23-42,
1968.
|
| 694. |
Salmoiraghi, G. C., and
B. D. Burns.
Localization and patterns of discharge of respiratory neurones in brain‐stem of cat.
J. Neurophysiol.
23:
1-26,
1960.
|
| 695. |
Saper, C. B.,
L. W. Swanson, and
W. M. Cowan.
The efferent connections of the ventromedial nucleus of the hypothalamus of the rat.
J. Comp. Neurol.
169:
409-442,
1976.
|
| 696. |
Sasa, M.,
S. Igarashi, and
S. Takaori.
Influence of the locus coeruleus in interneurons in the spinal trigeminal nucleus.
Brain Res.
125:
369-375,
1977.
|
| 697. |
Sasa, M., and
S. Takaori.
Influence of the locus coeruleus on transmission in the spinal trigeminal nucleus neurons.
Brain Res.
55:
203-208,
1973.
|
| 698. |
Sasaki, K.,
H. P. Staunton, and
G. Dieckmann.
Characteristic features of augmenting and recruiting responses in the cerebral cortex.
Exp. Neurol.
26:
369-392,
1970.
|
| 699. |
Sastre, J. P.,
K. Sakai, and
M. Jouvet.
Are the gigantocellular tegmental field neurons responsible for paradoxical sleep?
Brain Res.
229:
147-161,
1981.
|
| 700. |
Sastre, J. P.,
K. Sakai,
M. Tohyama, and
M. Jouvet.
Bilateral lesions of the dorso‐lateral pontine tegmentum. II. Effect upon muscle atonia (Abstract).
Sleep Res.
7:
44,
1978.
|
| 701. |
Sato, M.,
K. Itoh, and
N. Mizuno.
Distribution of thalamocaudal neurons in the cat as demonstrated by horseradish peroxidase.
Exp. Brain Res.
34:
143-153,
1979.
|
| 702. |
Satoh, K.,
M. Tohyama,
T. Yamamoto,
T. Sakumoto, and
N. Shimizu.
Noradrenaline innervation of the spinal cord studied by the horseradish peroxidase method combined with monoamine oxidase staining.
Exp. Brain Res.
30:
175-186,
1977.
|
| 703. |
Scheibel, A. B.
The problem of selective attention: a possible structural substrate. In:
Brain Mechanisms and Perceptual Awareness,
edited by O. Pompeiano and
C. Ajmone‐Marsan.
New York:
Raven,
1981,
p. 319-326.
|
| 704. |
Scheibel, M. E., and
A. B. Scheibel.
Structural substrates for integrative patterns in the brain stem reticular core. In:
Reticular Formation of the Brain,
edited by H. H. Jasper,
L. D. Proctor,
R. S. Knighton,
W. C. Noshay, and
R. T. Costello.
Boston, MA:
Little, Brown,
1958,
p. 31-55.
|
| 705. |
Scheibel, M. E., and
A. B. Scheibel.
Periodic sensory nonresponsiveness in reticular neurons.
Arch. Ital. Biol.
103:
300-316,
1965.
|
| 706. |
Scheibel, M. E., and
A. B. Scheibel.
The organization of the nucleus reticularis thalami: a Golgi study.
Brain Res.
1:
43-62,
1966.
|
| 707. |
Scheibel, M. E., and
A. B. Scheibel.
Structural organization of nonspecific thalamic nuclei and their projection toward cortex.
Brain Res.
6:
60-94,
1967.
|
| 708. |
Scheibel, M. E.,
A. B. Scheibel, and
T. Davis.
Some substrates for centrifugal control over thalamic cell ensembles. In:
Corticothalamic Projections and Sensorimotor Activities,
edited by T. Frigyesi,
E. Rinvik, and
M. Yahr.
New York:
Raven,
1972,
p. 131-156.
|
| 709. |
Scheibel, M.,
A. Scheibel,
A. Mollica, and
G. Moruzzi.
Convergence and interaction of afferent impulses on single units of reticular formation.
J. Neurophysiol.
18:
309-331,
1955.
|
| 710. |
Schlag, J. D., and
F. Chaillet.
Thalamic mechanisms involved in cortical desynchronization and recruiting responses.
Electroencephalogr. Clin. Neurophysiol.
15:
39-62,
1963.
|
| 711. |
Schlag, J.,
I. Lehtinen, and
M. Schlag‐Rey.
Neuronal activity before and during eye movements in thalamic internal medullary lamina of the cat.
J. Neurophysiol.
37:
982-995,
1974.
|
| 712. |
Schlag, J., and
J. Villablanca.
Cortical incremental responses to thalamic stimulation.
Brain Res.
6:
119-142,
1967.
|
| 713. |
Schlag, J., and
M. Waszak.
Electrophysiological properties of units of the thalamic reticular complex.
Exp. Neurol.
32:
79-97,
1971.
|
| 714. |
Schlauch, R.
Hypnopompic hallucinations and treatment with imipramine.
Am. J. Psychiatry
136:
219-220,
1979.
|
| 715. |
Schmielau, F.
Integration of visual and nonvisual information in nucleus reticularis thalami of the cat. In:
Developmental Neurobiology of Vision,
edited by R. D. Freeman.
New York:
Plenum,
1979,
p. 205-226.
|
| 716. |
Schwarcz, R.,
T. Hökfelt,
K. Fuxe,
G. Jonson,
M. Goldstein, and
L. Ternius.
Ibotenic acid‐induced neuronal degeneration: a morphobiological and neurochemical study.
Exp. Brain Res.
37:
199-216,
1979.
|
| 717. |
Schwartz, W. J.,
L. C. Davidsen, and
C. B. Smith.
In vivo metabolic activity of a putative circadian oscillator, the rat suprachiasmatic nucleus.
J. Comp. Neurol.
189:
157-167,
1980.
|
| 718. |
Schwartzkroin, P. A., and
C. E. Stafstrom.
Effects of EGTA on the calcium‐activated afterhyperpolarization in hippocampal CA3 pyramidal cells.
Science
210:
1125-1126,
1980.
|
| 719. |
Segal, M., and
F. E. Bloom.
The action of norepinephrine in the rat hippocampus. I. Iontophoretic studies.
Brain Res.
72:
79-97,
1974.
|
| 720. |
Segal, M., and
F. E. Bloom.
The action of norepinephrine in the rat hippocampus. II. Activation of the input pathway.
Brain Res.
72:
99-114,
1974.
|
| 721. |
Segal, M., and
F. E. Bloom.
The action of norepinephrine in the rat hippocampus. IV. The effects of locus coeruleus stimulation on evoked hippocampal unit activity.
Brain Res.
107:
513-525,
1976.
|
| 722. |
Segal, S., and
A. J. Mandell.
Behavioral activation of rats during intraventricular infusion of norepinephrine.
Proc. Natl. Acad. Sci. USA
66:
289-293,
1970.
|
| 723. |
Segundo, J. P.,
D. H. Perkel,
H. Wyman,
H. Hegstad, and
G. Moore.
Input‐output relations in computer‐simulated nerve cells.
Kybernetik
4:
157-171,
1968.
|
| 724. |
Segundo, J. P.,
T. Takenaka, and
H. Encabo.
Electro‐physiology of bulbar reticular neurons.
J. Neurophysiol.
30:
1194-1220,
1967.
|
| 725. |
Shaskan, E. G., and
S. H. Snyder.
Kinetics of serotonin accumulation into slices from rat brain: relationship to catecholamine uptake.
J. Pharmacol. Exp. Ther.
175:
404-418,
1970.
|
| 726. |
Sherrington, C.
Man on His Nature.
New York:
Doubleday,
1955.
|
| 727. |
Sheu, Y. S.,
J. P. Nelson, and
F. E. Bloom.
Discharge patterns of cat raphe neurons during sleep and waking.
Brain Res.
73:
263-276,
1974.
|
| 728. |
Shute, C. C. D., and
P. R. Lewis.
Cholinesterase containing systems of the brain of the rat.
Nature London
199:
1160-1164,
1963.
|
| 729. |
Shute, C. C. D., and
P. R. Lewis.
The ascending cholinergic reticular system: neocortical, olfactory and subcortical projections.
Brain
90:
497-520,
1967.
|
| 730. |
Sieck, G. C., and
R. M. Harper.
Pneumotaxic area neuronal discharge during sleep‐waking states in the cat.
Exp. Neurol.
67:
79-102,
1980.
|
| 731. |
Siegel, J. M.
Behavioral functions of the reticular formation.
Brain Res. Rev.
1:
69-105,
1979.
|
| 732. |
Siegel, J. M.,
D. G. McGinty, and
S. M. Breedlove.
Sleep and waking activity of pontine gigantocellular field neurons.
Exp. Neurol.
56:
553-573,
1977.
|
| 733. |
Siegel, J. M.,
R. L. Wheeler, and
D. J. McGinty.
Activity of medullary reticular formation neurons in the unrestrained cat during waking and sleep.
Brain Res.
179:
49-60,
1979.
|
| 734. |
Siegfried, B., and
J. Bures.
Asymmetry of EEG arousal in rats with unilateral 6‐hydroxydopamine lesions of substantia nigra: quantification of neglect.
Exp. Neurol.
62:
173-190,
1978.
|
| 735. |
Siggins, G. R.,
A. P. Oliver,
B. J. Hoffer, and
F. E. Bloom.
Cyclic adenosine monophosphate and norepinephrine effects on transmembrane properties of cerebellar Purkinje cells.
Science
171:
192-194,
1971.
|
| 736. |
Silberman, E. K.,
E. Vivaldi,
J. Garfield,
J. A. Hobson, and
R. W. McCarley.
Carbachol triggering of desynchronized sleep phenomena: enhancement via small volume infusions.
Brain Res.
191:
215-224,
1980.
|
| 737. |
Simonds, J. F.
Enuresis. A brief survey of current thinking with respect to pathogenesis and management.
Clin. Pediatr.
16:
79-82,
1977.
|
| 738. |
Sims, K. B.,
D. L. Hoffman,
S. I. Said, and
E. A. Zimmerman.
Vasoactive intestinal polypeptide (VIP) in mouse and rat brain: an immunocytochemical study.
Brain Res.
186:
165-183,
1980.
|
| 739. |
Singer, W.
The effect of mesencephalic reticular stimulation on intracellular potentials of cat lateral geniculate neurons.
Brain Res.
61:
35-54,
1973.
|
| 740. |
Singer, W.
Control of thalamic transmission by corticofugal and ascending reticular pathways in the visual system.
Physiol. Rev.
57:
386-420,
1977.
|
| 741. |
Singer, W.
Central‐core control of visual cortex functions. In:
The Neurosciences: Fourth Study Program,
edited by F. O. Schmitt and
F. G. Worden.
Cambridge, MA:
MIT Press,
1979,
p. 1093-1110.
|
| 742. |
Singer, W.,
F. Tretter, and
M. Cynader.
The effect of reticular stimulation on spontaneous and evoked activity in the cat visual cortex.
Brain Res.
102:
71-90,
1976.
|
| 743. |
Siteram, N.,
W. B. Mendelson,
R. J. Wyatt, and
J. C. Gillin.
The time‐dependent induction of REM sleep and arousal by physostigmine infusion during normal human sleep.
Brain Res.
122:
562-567,
1977.
|
| 744. |
Siteram, N.,
A. M. Moore, and
J. C. Gillin.
Experimental acceleration and slowing of REM sleep ultradian rhythm by cholinergic agonist and antagonist.
Nature London
274:
490-492,
1978.
|
| 745. |
Skinner, J. E., and
D. B. Lindsley.
Electrophysiological and behavioral effects of blockade of the nonspecific thalamocortical system.
Brain Res.
6:
95-118,
1967.
|
| 746. |
Skrebitsky, V. G., and
I. N. Sharonova.
Reticular suppression of flash‐evoked IPSPs in visual cortex neurons.
Brain Res.
111:
67-78,
1976.
|
| 747. |
Snyder, F.,
J. A. Hobson,
D. F. Morrison, and
F. Goldfrank.
Changes in respiration, heart rate, and systolic blood pressure in human sleep.
J. Appl. Physiol.
19:
417-422,
1964.
|
| 748. |
Sofroniew, M. V., and
A. Weindl.
Identification of parvocellular vasopressin and neurophysin neurons in the suprachiasmatic nucleus of a variety of mammals including primates.
J. Comp. Neurol.
193:
659-675,
1980.
|
| 749. |
Sokoloff, L.,
M. Reivich,
C. Kennedy,
M. H. Desrosiers,
C. S. Patlak,
K. D. Pettigrew,
O. Sakurada, and
M. Shinohara.
The [14C]deoxyglucose method for the measurement of local cerebral glucose utilization: theory, procedure, and normal values in the conscious and anesthetized albino rat.
J. Neurochem.
28:
897-916,
1977.
|
| 750. |
Spencer, W. A., and
J. M. Brookhart.
Electrical patterns of augmenting and recruiting waves in the depths of the sensorimotor cortex of cat.
J. Neurophysiol.
24:
26-49,
1961.
|
| 751. |
Spencer, W. A., and
J. M. Brookhart.
A study of spontaneous spindle waves in sensorimotor cortex of cat.
J. Neurophysiol.
24:
50-65,
1961.
|
| 752. |
Sprague, J. M.,
M. Levitt,
K. Robson,
C. N. Lin,
E. Stellar, and
W. W. Chambers.
A neuro‐anatomical and behavioral analysis of the syndromes resulting from midbrain lemniscal and reticular lesions in the cat.
Arch. Ital. Biol.
101:
225-295,
1963.
|
| 753. |
Stein, D.,
M. Jouvet, and
J. F. Pujol.
Effects of α‐methyl‐p‐tyrosine upon cerebral amine metabolism and sleep states in the cat.
Brain Res.
72:
360-365,
1974.
|
| 754. |
Steinbusch, H. W. M.,
D. van der Kooy,
A. A. J. Verhofstad, and
A. Pellegrino.
Serotonergic and non‐serotonergic projections from the nucleus raphe dorsalis to the caudateputamen complex in the rat, studied by a combined immuno‐fluorescence and fluorescent retrograde axonal labeling technique.
Neurosci. Lett.
19:
137-142,
1980.
|
| 755. |
Stephan, F. K.,
K. J. Berkley, and
R. L. Moss.
Efferent connections of the rat suprachiasmatic nucleus.
Neuroscience
6:
2625-2641,
1981.
|
| 756. |
Stephan, F. K., and
A. A. Nunez.
Elimination of circadian rhythms in drinking, activity, sleep and temperature by isolation of the suprachiasmatic nuclei.
Behav. Biol.
20:
1-16,
1977.
|
| 757. |
Steriade, M.
The flash‐evoked afterdischarge.
Brain Res.
9:
169-212,
1968.
|
| 758. |
Steriade, M.
Ascending control of motor cortex responsiveness.
Electroencephalogr. Clin. Neurophysiol.
26:
25-40,
1969.
|
| 759. |
Steriade, M.
Physiologie des voies et des centres visuels.
Paris:
Masson,
1969.
|
| 760. |
Steriade, M.
Ascending control of thalamic and cortical responsiveness.
Int. Rev. Neurobiol.
12:
87-144,
1970.
|
| 761. |
Steriade, M.
Cortical inhibition during sleep and waking. In:
Mechanisms in Transmission of Signal for Conscious Behaviour,
edited by T. Desiraju.
Amsterdam:
Elsevier,
1976,
p. 209-248.
|
| 762. |
Steriade, M.
Cortical long‐axoned cells and putative interneurons during the sleep‐waking cycle.
Behav. Brain Sci.
1:
465-514,
1978.
|
| 763. |
Steriade, M.
State‐dependent changes in the activity of rostral reticular and thalamocortical elements.
Neurosci. Res. Program Bull.
18:
83-91,
1980.
|
| 764. |
Steriade, M.
Mechanisms underlying cortical activation: neuronal organization and properties of the midbrain reticular core and intralaminar thalamic nuclei. In:
Brain Mechanisms of Perceptual Awareness and Purposeful Behavior,
edited by O. Pompeiano and
C. Ajmone‐Marsan.
New York:
Raven,
1981,
p. 327-377.
|
| 765. |
Steriade, M.
Midbrain reticular discharge related to forebrain activation processes. In:
Regulatory Functions of the CNS,
edited by J. Szentágothai,
M. Palkovits, and
J. Hámori.
Oxford, UK:
Pergamon,
1981,
vol. 1,
p. 283-292.
(Adv. Physiol. Sci.)
|
| 766. |
Steriade, M.
Cellular mechanisms of wakefulness and slow‐wave sleep. In:
Sleep Mechanisms and Functions in Humans and Animals: An Evolutionary Perspective,
edited by A. Mayes.
Wokingham, UK:
Van Nostrand Reinhold,
1983,
p. 161-216.
|
| 767. |
Steriade, M.
The excitatory‐inhibitory response sequence of thalamic and neocortical cells: state‐related changes and regulatory systems. In:
Dynamic Aspects of Neocortical Function,
edited by G. M. Edelman,
W. E. Gall, and
W. M. Cowan.
New York:
Wiley,
1984,
p. 107-157.
|
| 768. |
Steriade, M.,
V. Apostol, and
G. Oakson.
Control of unitary activities in cerebellothalamic pathway during wakefulness and synchronized sleep.
J. Neurophysiol.
34:
389-413,
1971.
|
| 769. |
Steriade, M.,
M. Belekhova, and
V. Apostol.
Reticular potentiation of cortical flash‐evoked afterdischarge.
Brain Res.
11:
276-280,
1968.
|
| 770. |
Steriade, M., and
M. Demetrescu.
Unspecific systems of inhibition and facilitation of potentials evoked by intermittent light.
J. Neurophysiol.
23:
602-617,
1960.
|
| 771. |
Steriade, M., and
M. Deschénes.
Cortical interneurons during sleep and waking in freely moving primates.
Brain Res.
50:
192-199,
1973.
|
| 772. |
Steriade, M., and
M. Deschénes.
Inhibitory processes and interneuronal apparatus in motor cortex during sleep and waking. II. Recurrent and afferent inhibition of pyramidal tract neurons.
J. Neurophysiol.
37:
1093-1113,
1974.
|
| 773. |
Steriade, M., and
M. Deschénes.
The thalamus as a neuronal oscillator.
Brain Res. Rev.
8:
1-63,
1984.
|
| 774. |
Steriade, M.,
M. Deschénes,
L. Domich, and
C. Mulle.
Abolition of spindle oscillations in thalamic neurons disconnected from nucleus reticularis thalami.
J. Neurophysiol.
54:
1473-1497,
1985.
|
| 775. |
Steriade, M.,
M. Deschénes, and
G. Oakson.
Inhibitory processes and interneuronal apparatus in motor cortex during sleep and waking. I. Background firing and responsiveness of pyramidal tract neurons and interneurons.
J. Neurophysiol.
37:
1065-1092,
1974.
|
| 776. |
Steriade, M.,
M. Deschénes,
P. Wyzinski, and
J. Y. Halle.
Input‐output organization of the motor cortex during sleep and waking. In:
Basic Sleep Mechanisms,
edited by O. Petre‐Quadens and
J. Schlag.
New York:
Academic,
1974,
p. 144-200.
|
| 777. |
Steriade, M.,
A. Diallo,
G. Oakson, and
B. White‐Guay.
Some synaptic inputs and telencephalic projections of lateralis posterior thalamic neurons.
Brain Res.
131:
39-53,
1977.
|
| 778. |
Steriade, M.,
L. Domich, and
G. Oakson.
Reticularis thalami neurons revisited: activity changes during shifts in states of vigilance.
J. Neurosci.
6:
68-81,
1986.
|
| 779. |
Steriade, M., and
L. L. Glenn.
Neocortical and caudate projections of intralaminar thalamic neurons and their synaptic excitation from the midbrain reticular core.
J. Neurophysiol.
48:
352-371,
1982.
|
| 780. |
Steriade, M., and
J. A. Hobson.
Neuronal activity during the sleep‐waking cycle.
Prog. Neurobiol.
6:
155-376,
1976.
|
| 781. |
Steriade, M.,
G. Iosif, and
V. Apostol.
Responsiveness of thalamic and cortical motor relays during arousal and various states of sleep.
J. Neurophysiol.
32:
251-265,
1969.
|
| 782. |
Steriade, M.,
A. Kitsikis, and
G. Oakson.
Thalamic inputs and subcortical targets of cortical neurons in areas 5 and 7 of cat.
Exp. Neurol.
60:
420-442,
1978.
|
| 783. |
Steriade, M.,
A. Kitsikis, and
G. Oakson.
Excitatory‐inhibitory processes in parietal association neurons during reticular activation and sleep‐waking cycle.
Sleep
1:
339-355,
1979.
|
| 784. |
Steriade, M.,
A. Kitsikis, and
G. Oakson.
Selectively REM‐related increased firing rates of cortical association interneurons during sleep: possible implications for learning. In:
Brain Mechanisms in Memory and Learning: From the Single Neuron to Man,
edited by M. A. B. Brazier.
New York:
Raven,
1979,
p. 47-52.
|
| 785. |
Steriade, M., and
D. Morin.
Reticular influences on primary and augmenting responses in primary somatosensory cortex.
Brain Res.
205:
67-80,
1981.
|
| 786. |
Steriade, M.,
G. Oakson, and
A. Diallo.
Reticular influences on lateralis posterior thalamic neurons.
Brain Res.
131:
55-71,
1977.
|
| 787. |
Steriade, M.,
G. Oakson, and
A. Kitsikis.
Firing rates and patterns of output and nonoutput cells in cortical areas 5 and 7 during the sleep‐waking cycle.
Exp. Neurol.
60:
443-468,
1978.
|
| 788. |
Steriade, M.,
G. Oakson, and
N. Ropert.
Firing rates and patterns of midbrain reticular neurons during steady and transitional states of the sleep‐waking cycle.
Exp. Brain Res.
46:
37-51,
1982.
|
| 789. |
Steriade, M.,
A. Parent, and
J. Hada.
Thalamic projections of nucleus reticularis thalami of cat: a study by using retrograde transport of horseradish peroxidase and fluorescent tracers.
J. Comp. Neurol.
229:
531-547,
1984.
|
| 790. |
Steriade, M.,
A. Parent,
N. Ropert, and
A. Kitsikis.
Zona incerta and lateral hypothalamic afferents to the midbrain reticular core of cat—an HRP and electrophysiological study.
Brain Res.
238:
13-28,
1982.
|
| 791. |
Steriade, M.,
N. Ropert,
A. Kitsikis, and
G. Oakson.
Ascending activating neuronal networks in midbrain reticular core and related rostral systems. In:
The Reticular Formation Revisited,
edited by J. A. Hobson and
M. A. B. Brazier.
New York:
Raven,
1980,
p. 125-167.
|
| 792. |
Steriade, M.,
K. Sakai, and
M. Jouvet.
Bulbothalamic neurons related to thalamocortical activation processes during paradoxical sleep.
Exp. Brain Res.
54:
463-475,
1984.
|
| 793. |
Steriade, M., and
P. Wyzinski.
Cortically elicited activities in thalamic reticularis neurons.
Brain Res.
42:
514-520,
1972.
|
| 794. |
Steriade, M.,
P. Wyzinski, and
V. Apostol.
Corticofugal projections governing rhythmic activity. In:
Corticothalamic Projections and Sensorimotor Activities,
edited by T. Frigyesi,
E. Rinvik, and
M. Yahr.
New York:
Raven,
1972,
p. 221-272.
|
| 795. |
Steriade, M.,
P. Wyzinski, and
V. Apostol.
Differential synaptic reactivity of simple and complex pyramidal tract neurons at various levels of vigilance.
Exp. Brain Res.
17:
87-110,
1973.
|
| 796. |
Steriade, M.,
P. Wyzinski,
M. Deschénes, and
M. Guerin.
Disinhibiton during waking in motor cortex neuronal chains in cat and monkey.
Brain Res.
30:
211-217,
1971.
|
| 797. |
Sterman, M. B., and
C. D. Clemente.
Forebrain inhibitory mechanisms: sleep patterns induced by basal forebrain stimulation in the behaving cat.
Exp. Neurol.
6:
103-117,
1962.
|
| 798. |
Stern, W. C., and
P. J. Morgane.
Effects of α‐methyltyrosine on REM sleep and brain amine levels in the cat.
Biol. Psychiatry
6:
301-306,
1973.
|
| 799. |
Stern, W. C., and
P. J. Morgane.
Effect of reserpine on sleep and brain biogenic amine levels in the cat.
Psychopharmacologia
28:
275-286,
1973.
|
| 800. |
Stone, T. W.
Cholinergic mechanisms in the rat somatosensory cerebral cortex.
J. Physiol. London
225:
485-499,
1972.
|
| 801. |
Stone, T. W.
Amino acids as neurotransmitters of corticofugal neurones in the rat: a comparison of glutamate and aspartate.
Br. J. Pharmacol.
67:
545-551,
1979.
|
| 802. |
Stone, T. W.,
D. A. Taylor, and
F. E. Bloom.
Cyclic AMP and cyclic GMP may mediate opposite neuronal responses in the rat cerebral cortex.
Science
187:
845-847,
1975.
|
| 803. |
Stool, S. E.,
R. D. Eavey,
N. L. Stein, and
W. B. Sharrar.
The “chubby puffer” syndrome. Upper airway obstruction and obesity, with intermittent somnolence and cardiorespiratory embarrassment.
Clin. Pediatr.
16:
43-50,
1977.
|
| 804. |
Strohl, K. P.,
N. A. Saunders,
N. T. Feldman, and
M. Hallett.
Obstructive sleep apnea in family members.
N. Engl. J. Med.
299:
969-973,
1978.
|
| 805. |
Sturmwasser, F.
Neurophysiological aspects of rhythm. In:
The Neurosciences: A Study Program,
edited by G. C. Quarton,
T. Melnechuk, and
F. O. Schmitt.
New York:
Rockefeller Univ. Press,
1967,
p. 516-528.
|
| 806. |
Sugimoto, T.,
N. Mizuno, and
K. Itoh.
An autoradiographic study on the terminal distribution of cerebellothalamic fibers in the cat.
Brain Res.
215:
29-47,
1981.
|
| 807. |
Sweet, C. P., and
J. A. Hobson.
The effects of posterior hypothalamic lesions on behavioral and electrographic manifestations of sleep and waking in cat.
Arch. Ital. Biol.
106:
283-293,
1968.
|
| 808. |
Szabo, J.
Striatonigral and nigrostriatal connections.
Appl. Neurophysiol.
42:
9-12,
1979.
|
| 809. |
Szentágothai, J.
Basic circuitry of the neocortex.
Exp. Brain Res. Suppl.
1:
282-287,
1976.
|
| 810. |
Szentágothai, J., and
K. Rajkovits.
Der Hirnnervenanteil der Pyramidenbahn und der prämotorische Apparat motorisher Hirnnervenkerne.
Arch. Psyciatr. Nervenkr.
197:
335-354,
1958.
|
| 811. |
Szerb, J. C.
Cortical acetylcholine release and electroencephalographic arousal.
J. Physiol. London
192:
329-345,
1967.
|
| 812. |
Taber, E.
The cytoarchitecture of the brain stem of the cat. I. Brain stem nuclei of the cat.
J. Comp. Neurol.
116:
27-70,
1961.
|
| 813. |
Taber, E.,
A. Brodal, and
F. Walberg.
The raphe nuclei of the brain stem in the cat. I. Normal topography and cytoarchitecture and general discussion.
J. Comp. Neurol.
114:
161-188,
1960.
|
| 814. |
Taber‐Pierce, E.,
W. F. Foote, and
J. A. Hobson.
The efferent connection of the nucleus raphe dorsalis.
Brain Res.
107:
137-144,
1976.
|
| 815. |
Thatcher, R. W., and
D. P. Purpura.
Postnatal development of thalamic synaptic events underlying evoked recruiting responses and electrocortical activation.
Brain Res.
60:
21-34,
1973.
|
| 816. |
Tissot, R.
The effects of certain drugs on the sleep cycle in man. In:
Progress in Brain Research. Sleep Mechanisms,
edited by K. Akert,
C. Bally, and
J. P. Schadé.
Amsterdam:
Elsevier,
1965,
vol. 18,
p. 175-177.
|
| 817. |
Tohyama, M.,
K. Sakai,
D. Salvert,
M. Touret, and
M. Jouvet.
Spinal projections from the lower brain stem in the cat as demonstrated by the horseradish peroxidase technique. I. Origins of the reticulospinal tracts and their funicular trajectories.
Brain Res.
173:
383-403,
1979.
|
| 818. |
Tömböl, T.
Collateral axonal arborization. In:
Golgi Centennial Symposium: Perspectives in Neurobiology,
edited by M. Santini.
New York:
Raven,
1975,
p. 133-141.
|
| 819. |
Törk, I., and
S. Turner.
Histochemical evidence for a cate‐cholaminergic (presumably dopaminergic) projection from the ventral mesencephalic tegmentum to visual cortex in the cat.
Neurosci. Lett.
24:
215-219,
1981.
|
| 820. |
Trulson, M. E., and
B. L. Jacobs.
Raphe unit activity in freely moving cats: correlation with level of behavioral arousal.
Brain Res.
163:
135-150,
1979.
|
| 821. |
Trulson, M. E.,
D. W. Preussler, and
G. A. Howell.
Activity of substantia nigra across the sleep‐waking cycle in freely moving cats.
Neurosci. Lett.
26:
183-188,
1981.
|
| 822. |
Udenfriend, S.,
P. Zaltzman‐Nirenberg,
R. Gordon, and
S. Spector.
Evaluation of the biochemical effects produced in vivo by inhibitors of the three enzymes involved in norepinephrine biosynthesis.
Mol. Pharmacol.
2:
95-105,
1966.
|
| 823. |
Valverde, F.
Reticular formation of the pons and medulla oblongata.
J. Comp. Neurol.
116:
71-99,
1961.
|
| 824. |
Valverde, F.
Reticular formation of the albino rat's brain stem: cytoarchitecture and corticofugal connections.
J. Comp. Neurol.
119:
25-53,
1962.
|
| 825. |
Van Buren, J. M., and
R. C. Borke.
Variations and Connections of the Human Thalamus.
New York:
Springer‐Verlag,
1972,
vol. 1.
|
| 826. |
Vanderwolf, C. H., and
T. E. Robinson.
Reticulo‐cortical activity and behavior: a critique of the arousal theory and a new synthesis.
Behav. Brain Sci.
4:
459-514,
1981.
|
| 827. |
Vandesande, F.,
K. Dierickx, and
J. De Mey.
Identification of the vasopressin‐neurophysin producing neurons of the rat suprachiasmatic nuclei.
Cell Tissue Res.
156:
377-380,
1975.
|
| 828. |
Van Dongen, P. A. M.
Locus coeruleus region: effects on behavior of cholinergic, noradrenergic, and opiate drugs injected intracerebrally into freely moving cats.
Exp. Neurol.
67:
52-78,
1980.
|
| 829. |
Vaughan, T.,
R. J. Wyatt, and
R. Green.
Changes in REM sleep of chronically anxious depressed patients given alpha‐methylparatyrosine (Abstract).
Psychophysiology
9:
197,
1971.
|
| 830. |
Velayos, J. L., and
F. Reinoso‐Suarez.
Topographic organization of the brainstem afferents to the mediodorsal thalamic nucleus.
J. Comp. Neurol.
206:
17-27,
1982.
|
| 831. |
Vertes, R. P.
Selective firing of rat pontine gigantocellular neurons during movement and REM sleep.
Brain Res.
128:
146-152,
1977.
|
| 832. |
Vertes, R. P.
Brain stem gigantocellular neurons: patterns of activity during behavior and sleep in the freely moving rat.
J. Neurophysiol.
42:
214-228,
1979.
|
| 833. |
Vibert, J. F.,
D. Caille,
F. Bertrand,
H. Gromysz, and
A. Hugelin.
Ascending projection from the respiratory centre to mesencephalon and diencephalon.
Neurosci. Lett.
11:
29-33,
1979.
|
| 834. |
Villablanca, J.
The electrocorticogram in the chronic cerveau isolé cat.
Electroencephalogr. Clin. Neurophysiol.
19:
576-586,
1965.
|
| 835. |
Vivaldi, E., and
J. A. Hobson.
Cholinergic REM sleep induction by the muscarinic agonist dioxolane (Abstract).
Sleep Res.
10:
104,
1981.
|
| 836. |
Vivaldi, E.,
R. W. McCarley, and
J. A. Hobson.
Evocation of desynchronized sleep signs by chemical microstimulation of the pontine brain stem. In:
The Reticular Formation Revisited,
edited by J. A. Hobson and
M. A. B. Brazier.
New York:
Raven,
1979,
p. 513-529.
|
| 837. |
Vogt, B. A.,
D. L. Rosene, and
D. N. Pandya.
Thalamic and cortical afferents differentiate anterior from posterior cingulate cortex in the monkey.
Science
204:
205-207,
1979.
|
| 838. |
Volterra, V.
Leçons sur la théorie mathématique de la lutte pour la vie.
Paris:
Gauthier‐Villars,
1931.
|
| 839. |
Walker, A. E.
The Primate Thalamus.
Chicago, IL:
Univ. of Chicago Press,
1938.
|
| 840. |
Wallach, M. B.,
W. D. Winters,
A. J. Mandell, and
C. E. Spooner.
A correlation of EEG, reticular multiple unit activity and gross behavior following various antidepressant agents in the cat.
Electroencephalogr. Clin. Neurophysiol.
27:
563-573,
1969.
|
| 841. |
Wallach, M. B.,
W. D. Winters,
A. J. Mandell, and
C. E. Spooner.
Effects of antidepressant drugs on wakefulness and sleep in the cat.
Electroencephalogr. Clin. Neurophysiol.
27:
574-580,
1969.
|
| 842. |
Walsh, J. T., and
J. P. Cordeau.
Responsiveness in the visual system during various phases of sleep and waking.
Exp. Neurol.
11:
90-103,
1965.
|
| 843. |
Walter, W. G.,
R. Cooper,
V. J. Aldridge,
W. C. McCallum, and
A. L. Winter.
Contingent negative variation: an electric sign of sensorimotor association and expectancy in the human brain.
Nature London
203:
380-384,
1964.
|
| 844. |
Waltzer, R. P., and
G. F. Martin.
A double labelling study demonstrating that most spinally projecting neurons of the nucleus reticularis gigantocellularis do not provide collateral innervation to the diencephalon in the rat.
Soc. Neurosci. Abstr.
9:
284,
1983.
|
| 845. |
Wang, R. Y., and
G. K. Aghajanian.
Antidromically identified serotonergic neurons in the rat midbrain raphe: evidence for collateral inhibition.
Brain Res.
132:
186-193,
1977.
|
| 846. |
Wang, R. Y., and
G. K. Aghajanian.
Physiological evidence for habenula as major link between forebrain and midbrain raphe.
Science
197:
89-91,
1977.
|
| 847. |
Wang, R. Y., and
G. K. Aghajanian.
Correlative firing patterns of serotonergic neurons in rat dorsal raphe nucleus.
J. Neurosci.
2:
11-16,
1982.
|
| 848. |
Wang, R. Y.,
D. W. Gallagher, and
G. K. Aghajanian.
Stimulation of pontine reticular formation suppresses firing of serotonergic neurones in the dorsal raphe.
Nature London
264:
365-368,
1976.
|
| 849. |
Wassef, M.,
A. Berod, and
C. Sotelo.
Dopaminergic dendrites in the pars reticulata of the rat substantia nigra and their striatal input. Combined immunocytochemical localization of tyrosine hydroxylase and anterograde degeneration.
Neuroscience
6:
2125-2139,
1981.
|
| 850. |
Waterhouse, B. D., and
D. J. Woodward.
Interaction of norepinephrine with cerebrocortical activity evoked by stimulation of somatosensory afferent pathways in the rat.
Exp. Neurol.
67:
11-34,
1980.
|
| 851. |
Watson, R. T.,
K. M. Heilman,
B. D. Miller, and
F. A. King.
Neglect after mesencephalic reticular formation lesions.
Neurology
24:
294-298,
1974.
|
| 852. |
Weiss, K. R., and
I. Kupfermann.
Serotonergic neuronal activity and arousal of feeding in Aplysia californica. In:
Aspects of Behavioral Neurobiology,
edited by J. A. Ferrendelli.
Washington, DC:
Soc. Neurosci.,
1977,
vol. 3,
p. 66-89.
(Soc. Neurosci. Symp.)
|
| 853. |
Weitzman, E. D.
Sleep‐wake, neuroendocrine and body temperature circadian rhythms under entrained and non‐entrained (free‐running) conditions in man. In:
Biological Rhythms and Their Central Mechanism,
edited by M. Suda,
O. Hayaishi, and
H. Nakagawa.
Amsterdam:
Elsevier/North‐Holland,
1979,
p. 199-227.
|
| 854. |
Werner, G., and
V. B. Mountcastle.
The variability of central neural activity in a sensory system and its implications for the central reflection of sensory events.
J. Neurophysiol.
26:
958-977,
1963.
|
| 855. |
West, C. H. K.,
J. C. Jackson, and
R. M. Benjamin.
An autoradiographic study of subcortical forebrain projections from mediodorsal and adjacent midline thalamic nuclei in the rabbit.
Neuroscience
4:
1977-1988,
1979.
|
| 856. |
Westman, J., and
D. Bowsher.
Fine structure of the centromedian‐parafascicular complex in the cat.
Brain Res.
30:
331-337,
1971.
|
| 857. |
Wiklund, L.,
L. Leger, and
M. Persson.
Monamine cell distribution in the cat brain stem. A fluorescence histochemical study with quantification of indolaminergic and locus coeruleus cell groups.
J. Comp. Neurol.
203:
613-647,
1981.
|
| 858. |
Williams, J. T.,
R. A. North,
S. A. Shefner,
S. Nishi, and
T. M. Egan.
Membrane properties of rat locus coeruleus neurones.
Neuroscience
13:
137-157,
1984.
|
| 859. |
Wilson, C. J.,
P. M. Groves, and
E. Fifkova.
Monoaminergic synapses, including dendro‐dendritic synapses in the rat substantia nigra.
Exp. Brain Res.
30:
161-174,
1977.
|
| 860. |
Woody, C. D.
Aspects of the electrophysiology of cortical processes related to the development and performance of learned motor responses.
Physiologist
17:
49-69,
1974.
|
| 861. |
Woody, C. D.,
B. E. Swartz, and
E. Gruen.
Effects of acetylcholine and cyclic GMP on input resistance of cortical neurons in awake cats.
Brain Res.
158:
373-395,
1978.
|
| 862. |
Wright, J. J., and
M. D. Craggs.
Changed cortical activation and lateral hypothalamic syndrome: a study in the split‐brain cat.
Brain Res.
151:
632-636,
1978.
|
| 863. |
Wuerthell, S. M.,
K. L. Lovell,
M. Z. Jones, and
K. E. Moore.
A histological study of kainic acid‐induced lesions in the rat brain.
Brain Res.
149:
489-497,
1978.
|
| 864. |
Wurtz, R. H.,
B. J. Richmond, and
W. T. Newsome.
Modulation of cortical visual processing by attention, perception, and movement. In:
Dynamic Aspects of Neocortical Function,
edited by G. M. Edelman,
W. E. Gall, and
W. M. Cowan.
New York:
Wiley,
1984,
p. 195-217.
|
| 865. |
Wyatt, R. J.
The serotonin‐catecholamine‐dream bicycle: a clinical study.
Biol. Psychiatry
5:
33-64,
1972.
|
| 866. |
Wyatt, R. J.,
T. N. Chase,
D. J. Kupfer,
J. Scott,
F. Snyder,
A. Sjoerdsma, and
K. Engelman.
Brain catecholamines and human sleep.
Nature London
233:
63-65,
1971.
|
| 867. |
Wyatt, R.,
D. J. Kupfer,
J. Scott,
D. S. Robinson, and
F. Snyder.
Longitudinal studies on the effects of monoamine oxidase inhibitors on sleep in man.
Psychopharmacologia
15:
236-244,
1969.
|
| 868. |
Wyzinski, P. W.,
R. W. McCarley, and
J. A. Hobson.
Discharge properties of pontine reticulospinal neurons during sleep‐waking cycle.
J. Neurophysiol.
41:
821-834,
1978.
|
| 869. |
Yamamoto, M., and
H. Nakahama.
Stochastic properties of spontaneous unit discharges in somatosensory cortex and mesencephalic reticular formation during sleep‐waking states.
J. Neurophysiol.
49:
1182-1198,
1983.
|
| 870. |
Yamoaka, S.
Participation of limbic‐hypothalamic structures in circadian rhythm of slow wave sleep and paradoxical sleep in the rat.
Brain Res.
151:
255-268,
1978.
|
| 871. |
Yen, C. T., and
E. G. Jones.
Intracellular staining of physiologically identified neurons and axons in the somatosensory thalamus of the cat.
Brain Res.
280:
148-154,
1983.
|
| 872. |
Yingling, C. D., and
J. E. Skinner.
Selective regulation of thalamic sensory relay nuclei by nucleus reticularis thalami. Electroencephalogr.
Clin. Neurophysiol.
41:
476-482,
1976.
|
| 873. |
Zarcone, V.
Narcolepsy.
N. Engl. J. Med.
288:
1156-1166,
1973.
|
| 874. |
Zatz, M., and
M. J. Brownstein.
Intraventricular carbachol mimics the effects of light on the circadian rhythm in the rat pineal gland.
Science
203:
358-361,
1979.
|
| 875. |
Zatz, M., and
M. J. Brownstein.
Injection of alpha‐bungarotoxin near the suprachiasmatic nucleus blocks the effects of light on nocturnal pineal enzyme activity.
Brain Res.
213:
438-442,
1981.
|
| 876. |
Zepelin, H., and
A. Rechtschaffen.
Mammalian sleep longevity and energy metabolism.
Brain Behav. Evol.
10:
425-470,
1974.
|