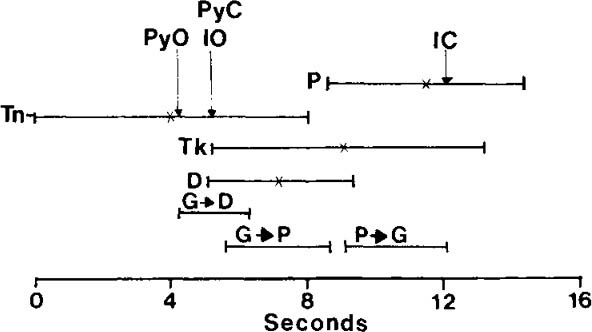
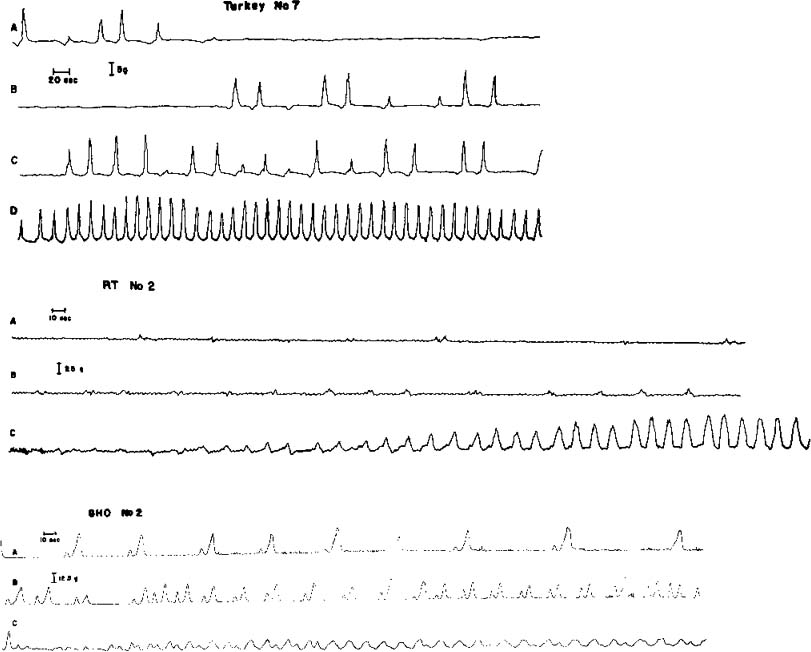
| References |
| 1. |
Ahmad, A.,
R. C. P. Singh, and
B. D. Garg.
Evidence of non‐cholinergic excitatory nervous transmission in chick ileum.
Life Sci.
22:
1049-1058,
1978.
|
| 2. |
Akester, A. R.,
R. S. Anderson,
K. J. Hill, and
G. W. Osbaldiston.
A radiographic study of urine flow in the domestic fowl.
Br. Poult Sci.
8:
209-212,
1967.
|
| 3. |
Aylott, M. V.,
O. H. Vestad,
J. F. Stephens, and
D. E. Turk.
Effect of coccidial infection upon passage rates of digestive tract contents of chicks.
Poult. Sci.
46:
900-904,
1968.
|
| 4. |
Balgooyen, T. G.
Pellet regurgitation of captive sparrow hawks.
Condor
73:
382-385,
1971.
|
| 5. |
Bartlet, A. L., and
T. Hassen.
Contraction of chicken rectum to nerve stimulation after blockade of sympathetic and parasympathetic transmission.
Q. J. Exp. Physiol. Cogn. Med. Sci.
56:
178-183,
1971.
|
| 6. |
Bennett, T.
The effects of hyoscine and anticholinesterases on cholinergic transmission to the smooth muscle cells of the avian gizzard.
Br. J. Pharmacol.
37:
585-594,
1969.
|
| 7. |
Bennett, T.
Studies on avian gizzard. Histochemical analysis of extrinsic and intrinsic innervation.
Z. Zellforsch. Mikrosk. Anat.
98:
188-201,
1969.
|
| 8. |
Bennett, T.
Nerve mediated excitation and inhibition of the smooth muscle cells of avian gizzard.
J. Physiol. Lond.
204:
669-686,
1969.
|
| 9. |
Bennett, T.
Peripheral and autonomic nervous systems. In:
Avian Biology,
edited by
D. S. Farner and
J. R. King.
London:
Academic,
1974,
vol. IV,
chapt. 13,
p. 1-77.
|
| 10. |
Bennett, T., and
J. L. S. Cobb.
Studies on avian gizzard morphology and innervation of smooth muscle.
Z. Zellforsch. Mikrosk. Anat.
96:
173-185,
1969.
|
| 11. |
Bennett, T., and
J. L. S. Cobb.
Studies on avian gizzard: Auerbach's plexus.
Z. Zellforsch. Mikrosk. Anat.
99:
109-120,
1969.
|
| 12. |
Bennett, T., and
J. Malmfors.
The adrenergic nervous system of domestic fowl (Gallus domesticus L.).
Z. Zellforsch. Mikrosk. Anat.
106:
22-50,
1970.
|
| 13. |
Bolton, T. B.
Physiology of nervous system. In:
Physiology and Biochemistry of Fowl,
edited by
D. J. Bell and
B. M. Freeman.
London:
Academic,
1971,
vol. 2,
chapt. 13,
p. 675-705.
|
| 14. |
Bortoff, A.
Digestion: motility.
Annu. Rev. Physiol.
34:
261-290,
1972.
|
| 15. |
Branch, J., and
J. H. Cummings.
Comparison of radio‐opaque pellets and chromium sesquioxide as inert markers in studies requiring accurate fecal collections.
Gut
19:
371-376,
1978.
|
| 16. |
Burnstock, C.
Evolution of the autonomic innervation of visceral and cardiovascular systems in vertebrates.
Pharmacol. Rev.
21:
247-324,
1969.
|
| 17. |
Calhoun, M.
Microscopic Anatomy of the Digestive System.
Ames:
Iowa State Univ. Press,
1954,
p. 1-127.
|
| 18. |
Chitty, D.
Pellet formation in short‐eared owls, Asio flammeus.
Proc. Zool. Soc. Lond.
108
(Series A):
267-287,
1938.
|
| 19. |
Chodnik, K. S.
Cytology of the glands associated with the alimentary tract of the domestic fowl (Gallus domesticus).
Q. J. Microsc. Sci.
89:
75-87,
1948.
|
| 20. |
Christensen, J.,
S. Anuras, and
R. L. Hauser.
Migrating spike bursts and electrical slow waves in the cat colon. Effect of sectioning.
Gastroenterology
66:
240-246,
1974.
|
| 21. |
Daniel, E. E.
Digestion: motor function.
Annu. Rev. Physiol.
31:
203-226,
1969.
|
| 22. |
Dansky, L. M., and
F. W. Hill.
Application of the chromic oxide indicator method to balance studies with growing chickens.
J. Nutr.
47:
449-459,
1952.
|
| 23. |
Duke, G. E.
Gastrointestinal motility and its regulation.
Poult. Sci.
61:
1245-1256,
1982.
|
| 24. |
Duke, G. E.
Avian digestion. In:
Duke's Physiology of Domestic Animals
(10th ed.),
edited by
M. J. Swenson
Ithaca, NY:
Cornell Univ. Press,
1983,
p. 359-366.
|
| 25. |
Duke, G. E.
Alimentary canal: anatomy, regulation of feeding and motility. In:
Avian Physiology
(4th ed.),
edited by
P. D. Sturkie
New York:
Springer‐Verlag,
1986,
chapt. 13,
p. 269-288.
|
| 26. |
Duke, G. E.
Raptor physiology. In:
Zoo and Wild Animal Medicine
(2nd ed.),
edited by
M. E. Fowler
Philadelphia, PA:
Saunders,
1985,
chapt. 13,
p. 370-376.
|
| 27. |
Duke, G. E.,
H. E. Dziuk, and
O. A. Evanson.
Gastric pressure and smooth muscle electrical potential changes in turkeys.
Am. J. Physiol.
222:
167-173,
1972.
|
| 28. |
Duke, G. E.,
H. E. Dziuk, and
L. Hawkins.
Gastrointestinal transit times in normal and bluecomb turkeys.
Poult. Sci.
48:
835-842,
1969.
|
| 29. |
Duke, G. E., and
O. A. Evanson.
Inhibition of gastric motility by duodenal contents in turkeys.
Poult. Sci.
51:
1625-1636,
1972.
|
| 30. |
Duke, G. E., and
O. A. Evanson.
Diurnal cycles of gastric motility in normal and fasted turkeys.
Poult. Sci.
55:
1802-1807,
1976.
|
| 31. |
Duke, G. E., and
O. A. Evanson.
Gastroduodenal electrical potential changes and contractile activity in birds of prey (Abstract).
Federation Proc.
35:
303,
1976.
|
| 32. |
Duke, G. E.,
O. A. Evanson,
J. G. Ciganek,
J. F. Miskowiec, and
T. E. Kostuch.
Inhibition of gastric motility in turkeys by intraduodenal injections of amino acid solutions.
Poult. Sci.
51:
1749-1757,
1972.
|
| 33. |
Duke, G. E.,
O. A. Evanson, and
D. R. Epstein.
Coordination of cecal motility during cecal evacuation.
Poult. Sci.
62:
545-550,
1983.
|
| 34. |
Duke, G. E.,
O. A. Evanson, and
B. J. Huberty.
Electrical potential changes and contractile activity of the distal cecum of turkeys.
Poult. Sci.
59:
1925-1934,
1980.
|
| 35. |
Duke, G. E.,
O. A. Evanson, and
A. A. Jagers.
Meal to pellet intervals in 14 species of captive raptors.
Comp. Biochem. Physiol. A Comp. Physiol.
53:
1-6,
1976.
|
| 36. |
Duke, G. E.,
O. A. Evanson, and
P. T. Redig.
A cephalic influence on gastric motility upon seeing food in domestic turkeys, Great‐horned owls (Bubo virginianus) and red‐tailed hawks (Bueto jamaicensis).
Poult. Sci.
55:
2155-2165,
1976.
|
| 37. |
Duke, G. E.,
O. A. Evanson,
P. T. Redig, and
D. D. Rhoades.
Mechanism of pellet egestion in great‐horned owls (Bubo virginianus).
Am. J. Physiol.
231:
1824-1829,
1976.
|
| 38. |
Duke, G. E., and
D. D. Rhoades.
Factors affecting meal to pellet intervals in great‐horned owls (Bubo virginianus).
Comp. Biochem. Physiol. A Comp. Physiol.
56:
283-286,
1977.
|
| 39. |
Duke, G. E.,
M. R. Fuller, and
B. J. Huberty.
The influence of hunger on meal to pellet intervals in barred owls.
Comp. Biochem. Physiol. A Comp. Physiol.
66:
203-207,
1980.
|
| 40. |
Duke, G. E.,
J. R. Kimmel,
K. Durham,
H. G. Pollock,
R. Bertoy, and
D. Rains‐Epstein.
Release of avian pancreatic polypeptide by various intraluminal contents in the stomach, duodenum or ileum of turkeys.
Dig. Dis. Sci.
27:
782-786,
1982.
|
| 41. |
Duke, G. E.,
J. R. Kimmel,
H. P. Hunt, and
H. G. Pollock.
The influence of avian pancreatic polypeptide on gastric secretion and motility in laying hens.
Poult. Sci.
64:
1231-1235,
1985.
|
| 42. |
Duke, G. E.,
J. R. Kimmel,
P. T. Redig, and
H. G. Pollock.
Influence of exogenous avian pancreatic polypeptide on gastrointestinal motility of domestic turkeys.
Poult. Sci.
58:
239-246,
1979.
|
| 43. |
Duke, G. E.,
T. E. Kostuch, and
O. A. Evanson.
Gastroduodenal electrical activity in turkeys.
Am. J. Dig. Dis.
20:
1047-1058,
1975.
|
| 44. |
Duke, G. E.,
T. E. Kostuch, and
O. A. Evanson.
Electrical activity and intraluminal pressure changes in the lower small intestine of turkeys.
Am. J. Dig. Dis.
20:
1040-1046,
1975.
|
| 45. |
Duke, G. E.,
G. A. Petrides, and
R. K. Ringer.
Chromium‐51 in food metabolizability and passage rate studies with the ring‐necked pheasant.
Poult. Sci.
48:
1356-1364,
1968.
|
| 46. |
Durham, K.
The Mechanism and Regulation of Pellet Egestion in the Red‐Tailed Hawk (Buteo jamaicensis) and Related Gastrointestinal Contractile Activity.
St. Paul:
Univ. of Minnesota,
1983.
|
| 47. |
Dziuk, H. E.
Reverse flow of gastrointestinal contents in turkeys (Abstract).
Federation Proc.
30:
610,
1971.
|
| 48. |
Dziuk, H. E., and
G. E. Duke.
Cineradiographic studies of gastric motility in turkeys.
Am. J. Physiol.
222:
159-166,
1972.
|
| 49. |
Everett, S. D.
Pharmacological responses of the isolated innervated intestine of the chick.
Br. J. Pharmacol. Chemother.
33:
342-348,
1968.
|
| 50. |
Farner, D. S.
Digestion and the digestive system. In:
Biology and Comparative Physiology of Birds,
edited by
A. J. Marshall
London:
Academic,
1960,
vol. I,
p. 411-467.
|
| 51. |
Fuller, M. R., and
G. E. Duke.
Regulation of pellet egestion: the effects of multiple feedings on meal to pellet intervals in great‐horned owls.
Comp. Biochem. Physiol. A Comp. Physiol.
62:
439-444,
1978.
|
| 52. |
Fuller, M. R.,
G. E. Duke, and
D. L. Eskedahl.
Regulation of pellet egestion: the influence of feeding time and soundproof conditions on meal to pellet intervals of red‐tailed hawks.
Comp. Biochem. Physiol. A Comp. Physiol.
62:
433-438,
1978.
|
| 53. |
Gonalons, E.,
R. Rial, and
J. A. Tur.
Phenol red as indicator of digestive tract motility in chickens.
Poult. Sci.
61:
581-583,
1982.
|
| 54. |
Grimm, R. J., and
W. M. Whitehouse.
Pellet formation in a great‐horned owl: a roentgenographic study.
Auk
80:
301-306,
1963.
|
| 55. |
Groebbels, F.
Der Vogel, Erster Band: Atmungswelt und Nahrungswelt.
Berlin:
Verlag von Gebruder Borntraeger,
1932.
|
| 56. |
Hill, K. J., and
P. J. Strachan.
Recent advances in digestive physiology of the fowl. In:
Symp. Zool. Soc. Lond. No. 35,
edited by
M. Peaker.
London:
Academic,
1975,
p. 1-12.
|
| 57. |
Hillerman, J. P.,
F. H. Kratzer, and
W. O. Wilson.
Food passage through chickens and turkeys and some regulating factors.
Poult. Sci.
32:
332-335,
1953.
|
| 58. |
Hodgkiss, J. P.
Peristalsis and antiperistalsis in the chicken caecum are myogenic.
Q. J. Exp. Physiol. Cogn. Med. Sci.
69:
161-170,
1984.
|
| 59. |
Imabayashi, K.,
M. Kametaka, and
T. Hatano.
Studies on digestion in the domestic fowl.
Tohoku J. Agric. Res.
6:
99-11,
1955.
|
| 60. |
Jerrett, S. A., and
W. R. Goodge.
Evidence for amylase in avian salivary glands.
J. Morphol.
139:
27-46,
1973.
|
| 61. |
Kostuch, T. E., and
G. E. Duke.
Gastric motility in great‐horned owls.
Comp. Biochem. Physiol. A. Comp. Physiol.
51:
201-205,
1975.
|
| 62. |
Lai, H. C., and
G. E. Duke.
Colonic motility in domestic turkeys.
Am. J. Dig. Dis.
23:
673-681,
1978.
|
| 63. |
Larbier, M.,
N. C. Baptista, and
J. C. Blum.
Effect of diet composition on digestive transit and amino acid intestinal absorption in chickens.
Ann. Biol. Anim. Biochim. Biophys.
17:
597-603,
1977.
|
| 64. |
Ludwick, J. R., and
P. Bass.
Contractile and electric activity of the extrahepatic biliary tract and duodenum.
Surg. Gynecol. Obstet.
124:
536-546,
1967.
|
| 65. |
Macowan, M. M., and
H. E. Magee.
Observations on digestion and absorption in fowls.
Q. J. Exp. Physiol. Cogn. Med. Sci.
21:
275-280,
1932.
|
| 66. |
Malagelada, J. R.,
S. E. Carter,
M. L. Brown, and
G. L. Carlson.
Radiolabeled fiber, a physiologic marker for gastric emptying and intestinal transit of solids.
Dig. Dis. Sci.
25:
81-87,
1980.
|
| 67. |
Mangold, E.
Die Verdauung bei den Nutztieren.
Berlin:
Akademie,
1950,
p. 87-93.
|
| 68. |
Mateos, G. G.,
J. L. Sell, and
J. A. Eastwood.
Rate of food passage (transit time) as influenced by level of supplemental fat.
Poult. Sci.
61:
94-100,
1982.
|
| 69. |
McLelland, J.
Digestive system. In:
Form and Function in Birds,
edited by
A. S. King and
J. McLelland.
London:
Academic,
1979,
p. 69-181.
|
| 70. |
Nolf, P.
On the existence in the bird of a system of intrinsic fibers connecting the stomach to the small intestine (Abstract).
J. Physiol. Lond.
90:
53P-54P,
1937.
|
| 71. |
Nolf, P.
L'appareil nerveux de l'automatisme gastrique de l'oiseau. I. Essai d'analyse par la nicotine.
Arch. Int. Physiol. Biochim.
46:
1-85,
1938.
|
| 72. |
Nolf, P.
L'appareil nerveux de l'automatisme gastrique de l'oiseau. II. Etude des effects causés par une ou plusieurs sections de l'anneau nerveaux du gesier.
Arch. Int. Physiol. Biochim.
46:
441-559,
1938.
|
| 73. |
Ohashi, H.
An electrophysiological study of transmission from intramural excitory nerve to smooth muscle cells of the chicken oesophagus.
Jpn. J. Pharmacol.
21:
585-596,
1971.
|
| 74. |
Oshima, S.,
K. Shimada, and
T. Tonoue.
Radiotelemetric observations of the diurnal changes in respiration rate, heart rate and intestinal motility of domestic fowl.
Poult. Sci.
53:
503-507,
1975.
|
| 75. |
Pastea, E.,
A. Nicolau, and
J. Rosca.
Dynamics of the digestive tract in hens and ducks.
Acta Physiol. Hung.
33:
305-310,
1968.
|
| 76. |
Patterson, T. L.
Gastric movements in the pigeon with economy of animal material. Comparative studies. V.
J. Lab. Clin. Med.
12:
1003-1008,
1927.
|
| 77. |
Pintea, V.,
V. Jarubescu, and
M. Cotrut.
Contributiuni la studiul esofagului de gaina.
Lucr. Stiint.
1:
297-310,
1957
[Cited in McLelland (70).].
|
| 78. |
Polin, D.,
E. R. Wynosky,
M. Loukides, and
C. C. Porter.
A possible urinary back flow to ceca revealed by studies on chicks with artificial anus and fed amprolium‐C14 or thiamine‐C14.
Poult. Sci.
46:
89-94,
1967.
|
| 79. |
Rea, A. M.
Turkey vultures casting pellets.
Auk
90:
209-210,
1973.
|
| 80. |
Reed, C. I., and
B. P. Reed.
The mechanism of pellet formation in the Great‐horned owl (Bubo virginianus).
Science
68:
359-360,
1928.
|
| 81. |
Rhoades, D. D., and
G. E. Duke.
Cineradiographic studies of gastric motility in great‐horned owls (Bubo virginianus).
Condor
79:
328-334,
1977.
|
| 82. |
Roche, M., and
J. Decerprit.
Contrôles hormonal et nerveux de la motricité du tractus digestif de la poule.
Ann. Rech. Vet.
8:
25-40,
1977.
|
| 83. |
Rogers, F. T.
Contribution to the physiology of the stomach. XXXIX. The hunger mechanism of the pigeon and its relation to the central nervous system.
Am. J. Physiol.
41:
555-570,
1916.
|
| 84. |
Rouff, H. J., and
K. F. Sewing.
Die rolle der kropfs bei der steurung der magensaftsekretion von huhnern.
Naunyn‐Schmiedebergs Arch. Exp. Pathol. Pharmakol.
271:
142-148,
1971.
|
| 85. |
Russell, J., and
P. Bass.
Labeling and gastric emptying of gels in dogs (Abstract).
Federation Proc.
42:
759,
1983.
|
| 86. |
Savory, C. J.,
G. E. Duke, and
R. W. Bertoy.
Influence of intravenous injections of cholecystokinin on gastrointestinal motility in turkeys and domestic fowls.
Comp. Biochem. Physiol. A. Comp. Physiol.
70:
179-189,
1981.
|
| 87. |
Savory, C. J., and
M. J. Gentle.
Intravenous injections of cholecystokinin and caerulin suppress food intake in domestic fowls.
Experientia Basel
36:
1191-1197,
1980.
|
| 88. |
Sibbald, I. R.
Passage of feed through the adult rooster.
Poult. Sci.
58:
446-459,
1979.
|
| 89. |
Sturkie, P. D.
Alimentary canal: anatomy, prehension, deglutition, feeding, drinking, passage of ingesta and motility. In:
Avian Physiology
(3rd ed.),
edited by
P. D. Sturkie
New York:
Springer‐Verlag,
1976,
p. 185-195.
|
| 90. |
Suzuki, M., and
S. Nomura.
Electromyographic studies on the deglutition movement in the fowl.
Jpn. J. Vet. Sci.
37:
289-293,
1975.
|
| 91. |
Thornton, P. A.,
P. J. Schaible, and
L. F. Wolterink.
Intestinal transit and skeletal retention of radioactive strontium in the chick.
Poult. Sci.
35:
1055-1060,
1956.
|
| 92. |
Tuckey, R.,
B. E. March, and
J. Biely.
Diet and the rate of food passage in the growing chick.
Poult. Sci.
37:
786-792,
1958.
|
| 93. |
Uden, P.,
P. E. Colucci, and
P. J. Van Soest.
Investigation of chromium, cerium and cobalt as markers in digesta. Rate of passage studies.
J. Sci. Food Agric.
31:
625-629,
1980.
|
| 94. |
Vonk, H. H., and
N. Postma.
X‐ray studies on the movements of the hen's intestine.
Physiol. Comp. Oecol.
1:
15-23,
1949.
|
| 95. |
Webb, T. E., and
J. R. Colvin.
The composition, structure and mechanism of formation of the lining of the gizzard of the chicken.
Can. J. Biochem. Physiol.
42:
59-70,
1964.
|
| 96. |
White, S. S.
The Larynx of Gallus domesticus.
Liverpool: Univ. of Liverpool,
1970
PhD thesis. [Cited in: McLelland (70).].
|
| 97. |
Wilson, E. K.,
F. W. Pierson,
P. Y. Hester,
R. L. Adams, and
W. J. Stadelman.
The effects of high environmental temperature on feed passage time and performance traits of Pekin ducks.
Poult. Sci.
59:
2322-2330,
1980.
|
| 98. |
Yntema, C. L., and
W. S. Hammond.
Experiments on the origin and development of the sacral autonomic nerves in chick embryo.
J. Exp. Zool.
129:
375-381,
1952.
|
| 99. |
Ziswiler, V., and
D. S. Farner.
Digestion and digestive system. In:
Avian Biology,
edited by
D. S. Farner and
J. R. King.
London:
Academic,
1972,
vol. II,
chapt. 13,
p. 343-430.
|