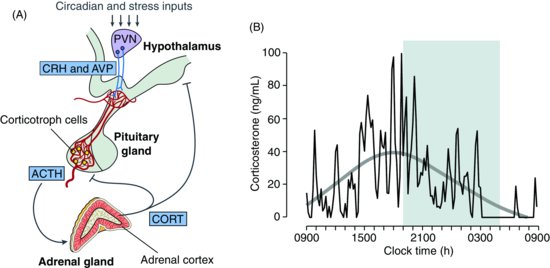
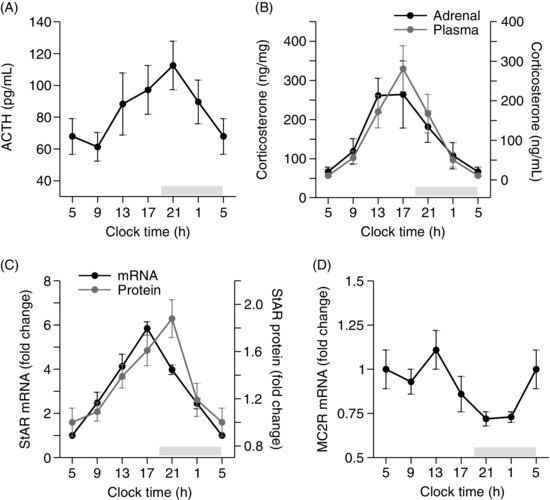
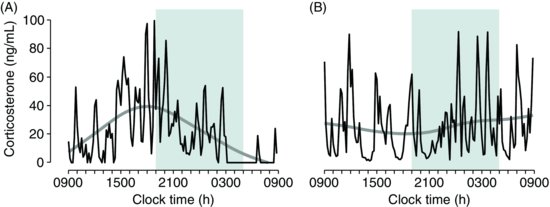
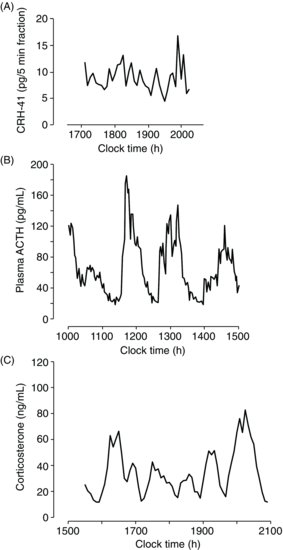
| References |
| 1. |
Abe
K
,
Kroning
J
,
Greer
MA
,
Critchlow
V
. Effects of destruction of the suprachiasmatic nuclei on the circadian rhythms in plasma corticosterone, body temperature, feeding and plasma thyrotropin. Neuroendocrinology
29: 119‐131, 1979. |
| 2. |
Aguilera
G
,
Jessop
DS
,
Harbuz
MS
,
Kiss
A
,
Lightman
SL
. Differential regulation of hypothalamic pituitary corticotropin releasing hormone receptors during development of adjuvant‐induced arthritis in the rat. J Endocrinol
153: 185‐191, 1997. |
| 3. |
Ahima
RS
,
Harlan
RE
. Charting of type II glucocorticoid receptor‐like immunoreactivity in the rat central nervous system. Neuroscience
39: 579‐604, 1990. |
| 4. |
Akana
SF
,
Cascio
CS
,
Du
JZ
,
Levin
N
,
Dallman
MF
. Reset of feedback in the adrenocortical system: An apparent shift in sensitivity of adrenocorticotropin to inhibition by corticosterone between morning and evening. Endocrinology
119: 2325‐2332, 1986. |
| 5. |
Alexander
LD
,
Sander
LD
. Vasoactive intestinal peptide stimulates ACTH and corticosterone release after injection into the PVN. Regul Pept
51: 221‐227, 1994. |
| 6. |
Antoni
FA
. Hypothalamic control of adrenocorticotropin secretion: Advances since the discovery of 41‐residue corticotropin‐releasing factor. Endoc Rev
7: 351‐378, 1986. |
| 7. |
Antoni
FA
,
Holmes
MC
,
Makara
GB
,
Karteszi
M
,
Laszlo
FA
. Evidence that the effects of arginine‐8‐vasopressin (AVP) on pituitary corticotropin (ACTH) release are mediated by a novel type of receptor. Peptides
5: 519‐522, 1984. |
| 8. |
Apostolakis
EM
,
Longo
LD
,
Veldhuis
JD
,
Yellon
SM
. Dissociation of pulsatile cortisol and adrenocorticotropin secretion in fetal sheep during late gestation. Endocrinology
130: 2571‐2578, 1992. |
| 9. |
Arakane
F
,
King
SR
,
Du
Y
,
Kallen
CB
,
Walsh
LP
,
Watari
H
,
Stocco
DM
,
Strauss
JF, II
. Phosphorylation of steroidogenic acute regulatory protein (StAR) modulates its steroidogenic activity. J Biol Chem
272: 32656‐32662, 1997. |
| 10. |
Arriza
JL
,
Simerly
RB
,
Swanson
LW
,
Evans
RM
. The neuronal mineralocorticoid receptor as a mediator of glucocorticoid response. Neuron
1: 887‐900, 1988. |
| 11. |
Artemenko
IP
,
Zhao
D
,
Hales
DB
,
Hales
KH
,
Jefcoate
CR
. Mitochondrial processing of newly synthesized steroidogenic acute regulatory protein (StAR), but not total StAR, mediates cholesterol transfer to cytochrome P450 side chain cleavage enzyme in adrenal cells. J Biol Chem
276: 46583‐46596, 2001. |
| 12. |
Ballard
PL
. Delivery and transport of glucocorticoids to target cells. Monogr Endocrinol
12: 25‐48, 1979. |
| 13. |
Bhake
R LJ
,
Linthorst
ACE
,
Lightman
SL
. Continuous ambulatory free cortisol profiles. In: Abstract of the 43rd Annual Meeting of the International Society of Psychoneuroendocrinology (ISPNE). Leiden, The Netherlands.: 2013. |
| 14. |
Bhake
RC
,
Leendertz
JA
,
Linthorst
AC
,
Lightman
SL
. Automated 24‐hours sampling of subcutaneous tissue free cortisol in humans. J Med Eng Technol
37: 180‐184, 2013. |
| 15. |
Bingaman
EW
,
Magnuson
DJ
,
Gray
TS
,
Handa
RJ
. Androgen inhibits the increases in hypothalamic corticotropin‐releasing hormone (CRH) and CRH‐immunoreactivity following gonadectomy. Neuroendocrinology
59: 228‐234, 1994. |
| 16. |
Bittman
EL
,
Doherty
L
,
Huang
L
,
Paroskie
A
. Period gene expression in mouse endocrine tissues. Am J Physiol Regul Integr Comp Physiol
285: R561‐R569, 2003. |
| 17. |
Buijs
RM
,
Kalsbeek
A
,
van der Woude
TP
,
van Heerikhuize
JJ
,
Shinn
S
. Suprachiasmatic nucleus lesion increases corticosterone secretion. Am J Physiol
264: R1186‐R1192, 1993. |
| 18. |
Buijs
RM
,
Van Eden
CG
. The integration of stress by the hypothalamus, amygdala and prefrontal cortex: Balance between the autonomic nervous system and the neuroendocrine system. Prog Brain Res
126: 117‐132, 2000. |
| 19. |
Buijs
RM
,
Wortel
J
,
Van Heerikhuize
JJ
,
Feenstra
MG
,
Ter Horst
GJ
,
Romijn
HJ
,
Kalsbeek
A
. Anatomical and functional demonstration of a multisynaptic suprachiasmatic nucleus adrenal (cortex) pathway. Eur J Neurosci
11: 1535‐1544, 1999. |
| 20. |
Bunger
MK
,
Wilsbacher
LD
,
Moran
SM
,
Clendenin
C
,
Radcliffe
LA
,
Hogenesch
JB
,
Simon
MC
,
Takahashi
JS
,
Bradfield
CA
. Mop3 is an essential component of the master circadian pacemaker in mammals. Cell
103: 1009‐1017, 2000. |
| 21. |
Bush
IE
. Species differences in adrenocortical secretion. J Endocrinol
9: 95‐100, 1953. |
| 22. |
Camacho‐Arroyo
I
,
Neri‐Gomez
T
,
Gonzalez‐Arenas
A
,
Guerra‐Araiza
C
. Changes in the content of steroid receptor coactivator‐1 and silencing mediator for retinoid and thyroid hormone receptors in the rat brain during the estrous cycle. J Steroid Biochem Mol Biol
94: 267‐272, 2005. |
| 23. |
Cameron
A
,
Henley
D
,
Carrell
R
,
Zhou
A
,
Clarke
A
,
Lightman
S
. Temperature‐responsive release of cortisol from its binding globulin: A protein thermocouple. J Clin Endocrinol Metab
95: 4689‐4695, 2010. |
| 24. |
Caraty
A
,
Grino
M
,
Locatelli
A
,
Oliver
C
. Secretion of corticotropin releasing factor (CRF) and vasopressin (AVP) into the hypophysial portal blood of conscious, unrestrained rams. Biochem Biophys Res Commun
155: 841‐849, 1988. |
| 25. |
Carnes
M
,
Brownfield
MS
,
Kalin
NH
,
Lent
S
,
Barksdale
CM
. Episodic secretion of ACTH in rats. Peptides
7: 219‐223, 1986. |
| 26. |
Carnes
M
,
Kalin
NH
,
Lent
SJ
,
Barksdale
CM
,
Brownfield
MS
. Pulsatile ACTH secretion: Variation with time of day and relationship to cortisol. Peptides
9: 325‐331, 1988. |
| 27. |
Carnes
M
,
Lent
S
,
Feyzi
J
,
Hazel
D
. Plasma adrenocorticotropic hormone in the rat demonstrates three different rhythms within 24 h. Neuroendocrinology
50: 17‐25, 1989. |
| 28. |
Carnes
M
,
Lent
SJ
,
Erisman
S
,
Barksdale
C
,
Feyzi
J
. Immunoneutralization of corticotropin‐releasing hormone prevents the diurnal surge of ACTH. Life Sci
45: 1049‐1056, 1989. |
| 29. |
Carnes
M
,
Lent
SJ
,
Erisman
S
,
Feyzi
J
. Changes in mean plasma ACTH reflect changes in amplitude and frequency of secretory pulses. Life Sci
43: 1785‐1790, 1988. |
| 30. |
Chalmers
DT
,
Lovenberg
TW
,
De Souza
EB
. Localization of novel corticotropin‐releasing factor receptor (CRF2) mRNA expression to specific subcortical nuclei in rat brain: Comparison with CRF1 receptor mRNA expression. J Neurosci
15: 6340‐6350, 1995. |
| 31. |
Chaudhary
LR
,
Stocco
DM
. Effect of different steroidogenic stimuli on protein phosphorylation and steroidogenesis in MA‐10 mouse Leydig tumor cells. Biochim Biophys Acta
1094: 175‐184, 1991. |
| 32. |
Cherrington
AD
. Banting Lecture 1997. Control of glucose uptake and release by the liver in vivo. Diabetes
48: 1198‐1214, 1999. |
| 33. |
Chikanza
IC
,
Chrousos
G
,
Panayi
GS
. Abnormal neuroendocrine immune communications in patients with rheumatoid arthritis. Eur J Clin Invest
22: 635‐637, 1992. |
| 34. |
Chowdrey
HS
,
Larsen
PJ
,
Harbuz
MS
,
Jessop
DS
,
Aguilera
G
,
Eckland
DJ
,
Lightman
SL
. Evidence for arginine vasopressin as the primary activator of the HPA axis during adjuvant‐induced arthritis. Br J Pharmacol
116: 2417‐2424, 1995. |
| 35. |
Chrousos
GP
. The hypothalamic‐pituitary‐adrenal axis and immune‐mediated inflammation. N Engl J Med
332: 1351‐1362, 1995. |
| 36. |
Churchill
PF
,
Kimura
T
. Topological studies of cytochromes P‐450scc and P‐45011 beta in bovine adrenocortical inner mitochondrial membranes. Effects of controlled tryptic digestion. J Biol Chem
254: 10443‐10448, 1979. |
| 37. |
Clark
RG
,
Chambers
G
,
Lewin
J
,
Robinson
IC
. Automated repetitive microsampling of blood: Growth hormone profiles in conscious male rats. J Endocrinol
111: 27‐35, 1986. |
| 38. |
Conway‐Campbell
BL
,
George
CL
,
Pooley
JR
,
Knight
DM
,
Norman
MR
,
Hager
GL
,
Lightman
SL
. The HSP90 molecular chaperone cycle regulates cyclical transcriptional dynamics of the glucocorticoid receptor and its coregulatory molecules CBP/p300 during ultradian ligand treatment. Mol Endocrinol
25: 944‐954, 2011. |
| 39. |
Conway‐Campbell
BL
,
McKenna
MA
,
Wiles
CC
,
Atkinson
HC
,
de Kloet
ER
,
Lightman
SL
. Proteasome‐dependent down‐regulation of activated nuclear hippocampal glucocorticoid receptors determines dynamic responses to corticosterone. Endocrinology
148: 5470‐5477, 2007. |
| 40. |
Conway‐Campbell
BL
,
Pooley
JR
,
Hager
GL
,
Lightman
SL
. Molecular dynamics of ultradian glucocorticoid receptor action. Mol Cell Endocrinol
348: 383‐393, 2012. |
| 41. |
Conway‐Campbell
BL
,
Sarabdjitsingh
RA
,
McKenna
MA
,
Pooley
JR
,
Kershaw
YM
,
Meijer
OC
,
De Kloet
ER
,
Lightman
SL
. Glucocorticoid ultradian rhythmicity directs cyclical gene pulsing of the clock gene period 1 in rat hippocampus. J Neuroendocrinol
22: 1093‐1100, 2010. |
| 42. |
Dallman
MF
,
Akana
SF
,
Cascio
CS
,
Darlington
DN
,
Jacobson
L
,
Levin
N
. Regulation of ACTH secretion: Variations on a theme of B. Recent Prog Horm Res
43: 113‐173, 1987. |
| 43. |
Dallman
MF
,
Akana
SF
,
Jacobson
L
,
Levin
N
,
Cascio
CS
,
Shinsako
J
. Characterization of corticosterone feedback regulation of ACTH secretion. Ann N Y Acad Sci
512: 402‐414, 1987. |
| 44. |
Dallman
MF
,
Engeland
WC
,
Rose
JC
,
Wilkinson
CW
,
Shinsako
J
,
Siedenburg
F
. Nycthemeral rhythm in adrenal responsiveness to ACTH. Am J Physiol
235: R210‐R218, 1978. |
| 45. |
Dallman
MF
,
Jones
MT
. Corticosteroid feedback control of ACTH secretion: Effect of stress‐induced corticosterone ssecretion on subsequent stress responses in the rat. Endocrinology
92: 1367‐1375, 1973. |
| 46. |
Dallmann
R
,
Touma
C
,
Palme
R
,
Albrecht
U
,
Steinlechner
S
. Impaired daily glucocorticoid rhythm in Per1 (Brd) mice. J Comp Physiol [A]
192: 769‐775, 2006. |
| 47. |
De Kloet
ER
. Hormones and the stressed brain. Ann N Y Acad Sci
1018: 1‐15, 2004. |
| 48. |
de Kloet
ER
. Steroids, stability and stress. Front Neuroendocrinol
16: 416‐425, 1995. |
| 49. |
de Kloet
ER
. Stress in the brain. Eur J Pharmacol
405: 187‐198, 2000. |
| 50. |
De Kloet
ER
,
Vreugdenhil
E
,
Oitzl
MS
,
Joels
M
. Brain corticosteroid receptor balance in health and disease. Endoc Rev
19: 269‐301, 1998. |
| 51. |
Di
S
,
Malcher‐Lopes
R
,
Halmos
KC
,
Tasker
JG
. Nongenomic glucocorticoid inhibition via endocannabinoid release in the hypothalamus: A fast feedback mechanism. J Neurosci
23: 4850‐4857, 2003. |
| 52. |
Droste
SK
,
Collins
A
,
Lightman
SL
,
Linthorst
AC
,
Reul
JM
. Distinct, time‐dependent effects of voluntary exercise on circadian and ultradian rhythms and stress responses of free corticosterone in the rat hippocampus. Endocrinology
150: 4170‐4179, 2009. |
| 53. |
Droste
SK
,
de Groote
L
,
Atkinson
HC
,
Lightman
SL
,
Reul
JM
,
Linthorst
AC
. Corticosterone levels in the brain show a distinct ultradian rhythm but a delayed response to forced swim stress. Endocrinology
149: 3244‐3253, 2008. |
| 54. |
Droste
SK
,
de Groote
L
,
Lightman
SL
,
Reul
JM
,
Linthorst
AC
. The ultradian and circadian rhythms of free corticosterone in the brain are not affected by gender: An in vivo microdialysis study in Wistar rats. J Neuroendocrinol
21: 132‐140, 2009. |
| 55. |
Engler
D
,
Pham
T
,
Fullerton
MJ
,
Clarke
IJ
,
Funder
JW
. Evidence for an ultradian secretion of adrenocorticotropin, beta‐endorphin and alpha‐melanocyte‐stimulating hormone by the ovine anterior and intermediate pituitary. Neuroendocrinology
49: 349‐360, 1989. |
| 56. |
Engler
D
,
Pham
T
,
Liu
JP
,
Fullerton
MJ
,
Clarke
IJ
,
Funder
JW
. Studies of the regulation of the hypothalamic‐pituitary‐adrenal axis in sheep with hypothalamic‐pituitary disconnection. II. Evidence for in vivo ultradian hypersecretion of proopiomelanocortin peptides by the isolated anterior and intermediate pituitary. Endocrinology
127: 1956‐1966, 1990. |
| 57. |
Evanson
NK
,
Tasker
JG
,
Hill
MN
,
Hillard
CJ
,
Herman
JP
. Fast feedback inhibition of the HPA axis by glucocorticoids is mediated by endocannabinoid signaling. Endocrinology
151: 4811‐4819, 2010. |
| 58. |
Fahrenkrug
J
,
Hannibal
J
,
Georg
B
. Diurnal rhythmicity of the canonical clock genes Per1, Per2 and Bmal1 in the rat adrenal gland is unaltered after hypophysectomy. J Neuroendocrinol
20: 323‐329, 2008. |
| 59. |
Fulkerson
WJ
. Synchronous episodic release of cortisol in the sheep. J Endocrinol
79: 131‐132, 1978. |
| 60. |
Gala
RR
,
Westphal
U
. Corticosteroid‐binding globulin in the rat: Studies on the sex difference. Endocrinology
77: 841‐851, 1965. |
| 61. |
Gekakis
N
,
Staknis
D
,
Nguyen
HB
,
Davis
FC
,
Wilsbacher
LD
,
King
DP
,
Takahashi
JS
,
Weitz
CJ
. Role of the CLOCK protein in the mammalian circadian mechanism. Science
280: 1564‐1569, 1998. |
| 62. |
Gillies
GE
,
Linton
EA
,
Lowry
PJ
. Corticotropin releasing activity of the new CRF is potentiated several times by vasopressin. Nature
299: 355‐357, 1982. |
| 63. |
Girotti
M
,
Pace
TW
,
Gaylord
RI
,
Rubin
BA
,
Herman
JP
,
Spencer
RL
. Habituation to repeated restraint stress is associated with lack of stress‐induced c‐fos expression in primary sensory processing areas of the rat brain. Neuroscience
138: 1067‐1081, 2006. |
| 64. |
Graham
ES
,
Littlewood
P
,
Turnbull
Y
,
Mercer
JG
,
Morgan
PJ
,
Barrett
P
. Neuromedin‐U is regulated by the circadian clock in the SCN of the mouse. Eur J Neurosci
21: 814‐819, 2005. |
| 65. |
Haller
J
,
Halasz
J
,
Mikics
E
,
Kruk
MR
,
Makara
GB
. Ultradian corticosterone rhythm and the propensity to behave aggressively in male rats. J Neuroendocrinol
12: 937‐940, 2000. |
| 66. |
Haller
J
,
Millar
S
,
van de Schraaf
J
,
de Kloet
RE
,
Kruk
MR
. The active phase‐related increase in corticosterone and aggression are linked. J Neuroendocrinol
12: 431‐436, 2000. |
| 67. |
Handa
RJ
,
Weiser
MJ
. Gonadal steroid hormones and the hypothalamo‐pituitary‐adrenal axis. Front Neuroendocrinol
35: 197‐220, 2014. |
| 68. |
Harbuz
MS
,
Rees
RG
,
Eckland
D
,
Jessop
DS
,
Brewerton
D
,
Lightman
SL
. Paradoxical responses of hypothalamic corticotropin‐releasing factor (CRF) messenger ribonucleic acid (mRNA) and CRF‐41 peptide and adenohypophysial proopiomelanocortin mRNA during chronic inflammatory stress. Endocrinology
130: 1394‐1400, 1992. |
| 69. |
Harbuz
MS
,
Rees
RG
,
Lightman
SL
. HPA axis responses to acute stress and adrenalectomy during adjuvant‐induced arthritis in the rat. Am J Physiol
264: R179‐R185, 1993. |
| 70. |
Hastings
M
,
Maywood
ES
. Circadian clocks in the mammalian brain. BioEssays
22: 23‐31, 2000. |
| 71. |
Hastings
M
,
O'Neill
JS
,
Maywood
ES
. Circadian clocks: Regulators of endocrine and metabolic rhythms. J Endocrinol
195: 187‐198, 2007. |
| 72. |
Hendrickson
AE
,
Wagoner
N
,
Cowan
WM
. An autoradiographic and electron microscopic study of retino‐hypothalamic connections. Z Zellforsch Mikrosk Anat
135: 1‐26, 1972. |
| 73. |
Henley
DE
,
Leendertz
JA
,
Russell
GM
,
Wood
SA
,
Taheri
S
,
Woltersdorf
WW
,
Lightman
SL
. Development of an automated blood sampling system for use in humans. J Med Eng Technol
33: 199‐208, 2009. |
| 74. |
Hill
MN
,
Tasker
JG
. Endocannabinoid signaling, glucocorticoid‐mediated negative feedback, and regulation of the hypothalamic‐pituitary‐adrenal axis. Neuroscience
204: 5‐16, 2012. |
| 75. |
Hinz
B
,
Hirschelmann
R
. Rapid non‐genomic feedback effects of glucocorticoids on CRF‐induced ACTH secretion in rats. Pharm Res
17: 1273‐1277, 2000. |
| 76. |
Holaday
JW
,
Martinez
HM
,
Natelson
BH
. Synchronized ultradian cortisol rhythms in monkeys: Persistence during corticotropin infusion. Science
198: 56‐58, 1977. |
| 77. |
Hsu
BR
,
Kuhn
RW
. The role of the adrenal in generating the diurnal variation in circulating levels of corticosteroid‐binding globulin in the rat. Endocrinology
122: 421‐426, 1988. |
| 78. |
Hubler
TR
,
Scammell
JG
. Intronic hormone response elements mediate regulation of FKBP5 by progestins and glucocorticoids. Cell Stress Chaperones
9: 243‐252, 2004. |
| 79. |
Hugin‐Flores
ME
,
Steimer
T
,
Aubert
ML
,
Schulz
P
. Mineralo‐ and glucocorticoid receptor mrnas are differently regulated by corticosterone in the rat hippocampus and anterior pituitary. Neuroendocrinology
79: 174‐184, 2004. |
| 80. |
Hum
DW
,
Miller
WL
. Transcriptional regulation of human genes for steroidogenic enzymes. Clin Chem
39: 333‐340, 1993. |
| 81. |
Ishida
A
,
Mutoh
T
,
Ueyama
T
,
Bando
H
,
Masubuchi
S
,
Nakahara
D
,
Tsujimoto
G
,
Okamura
H
. Light activates the adrenal gland: Timing of gene expression and glucocorticoid release. Cell Metab
2: 297‐307, 2005. |
| 82. |
Iwasaki‐Sekino
A
,
Mano‐Otagiri
A
,
Ohata
H
,
Yamauchi
N
,
Shibasaki
T
. Gender differences in corticotropin and corticosterone secretion and corticotropin‐releasing factor mRNA expression in the paraventricular nucleus of the hypothalamus and the central nucleus of the amygdala in response to footshock stress or psychological stress in rats. Psychoneuroendocrinology
34: 226‐237, 2009. |
| 83. |
Ixart
G
,
Barbanel
G
,
Nouguier‐Soule
J
,
Assenmacher
I
. A quantitative study of the pulsatile parameters of CRH‐41 secretion in unanesthetized free‐moving rats. Exp Brain Res
87: 153‐158, 1991. |
| 84. |
Ixart
G
,
Siaud
P
,
Barbanel
G
,
Mekaouche
M
,
Givalois
L
,
Assenmacher
I
. Circadian variations in the amplitude of corticotropin‐releasing hormone 41 (CRH41) episodic release measured in vivo in male rats: Correlations with diurnal fluctuations in hypothalamic and median eminence CRH41 contents. J Biol Rhythms
8: 297‐309, 1993. |
| 85. |
Ixart
G
,
Siaud
P
,
Mekaouche
M
,
Barbanel
G
,
Givalois
L
,
Assenmacher
I
. Short‐term but not long‐term adrenalectomy modulates amplitude and frequency of the CRH41 episodic release in push‐pull cannulated median eminence of free‐moving rats. Brain Res
658: 185‐191, 1994. |
| 86. |
Jasper
MS
,
Engeland
WC
. Synchronous ultradian rhythms in adrenocortical secretion detected by microdialysis in awake rats. Am J Physiol
261: R1257‐R1268, 1991. |
| 87. |
Jasper
MS
,
Engeland
WC
. Splanchnic neural activity modulates ultradian and circadian rhythms in adrenocortical secretion in awake rats. Neuroendocrinology
59: 97‐109, 1994. |
| 88. |
Jasper
MS
,
Engeland
WC
. Splanchnicotomy increases adrenal sensitivity to ACTH in nonstressed rats. Am J Physiol
273: E363‐E368, 1997. |
| 89. |
Joels
M
,
Urban
IJ
. The effect of microiontophoretically applied vasopressin and oxytocin on single neurones in the septum and dorsal hippocampus of the rat. Neurosci Lett
33: 79‐84, 1982. |
| 90. |
Jones
MT
,
Brush
FR
,
Neame
RL
. Characteristics of fast feedback control of corticotrophin release by corticosteroids. J Endocrinol
55: 489‐497, 1972. |
| 91. |
Jones
MT
,
Hillhouse
EW
,
Burden
JL
. Dynamics and mechanics of corticosteroid feedback at the hypothalamus and anterior pituitary gland. J Endocrinol
73: 405‐417, 1977. |
| 92. |
Jones
MT
,
Tiptaft
EM
,
Brush
FR
,
Fergusson
DA
,
Neame
RL
. Evidence for dual corticosteroid‐receptor mechanisms in the feedback control of adrenocorticotrophin secretion. J Endocrinol
60: 223‐233, 1974. |
| 93. |
Kalsbeek
A
,
Buijs
RM
,
van Heerikhuize
JJ
,
Arts
M
,
van der Woude
TP
. Vasopressin‐containing neurons of the suprachiasmatic nuclei inhibit corticosterone release. Brain Res
580: 62‐67, 1992. |
| 94. |
Kalsbeek
A
,
Fliers
E
,
Hofman
MA
,
Swaab
DF
,
Buijs
RM
. Vasopressin and the output of the hypothalamic biological clock. J Neuroendocrinol
22: 362‐372, 2010. |
| 95. |
Kalsbeek
A
,
van der Spek
R
,
Lei
J
,
Endert
E
,
Buijs
RM
,
Fliers
E
. Circadian rhythms in the hypothalamo‐pituitary‐adrenal (HPA) axis. Mol Cell Endocrinol
349: 20‐29, 2012. |
| 96. |
Kalsbeek
A
,
van der Vliet
J
,
Buijs
RM
. Decrease of endogenous vasopressin release necessary for expression of the circadian rise in plasma corticosterone: A reverse microdialysis study. J Neuroendocrinol
8: 299‐307, 1996. |
| 97. |
Kalsbeek
A
,
van Heerikhuize
JJ
,
Wortel
J
,
Buijs
RM
. A diurnal rhythm of stimulatory input to the hypothalamo‐pituitary‐adrenal system as revealed by timed intrahypothalamic administration of the vasopressin V1 antagonist. J Neurosci
16: 5555‐5565, 1996. |
| 98. |
Kaneko
M
,
Hiroshige
T
,
Shinsako
J
,
Dallman
MF
. Diurnal changes in amplification of hormone rhythms in the adrenocortical system. Am J Physiol
239: R309‐R316, 1980. |
| 99. |
Karssen
AM
,
Meijer
OC
,
van der Sandt
IC
,
Lucassen
PJ
,
de Lange
EC
,
de Boer
AG
,
de Kloet
ER
. Multidrug resistance P‐glycoprotein hampers the access of cortisol but not of corticosterone to mouse and human brain. Endocrinology
142: 2686‐2694, 2001. |
| 100. |
Karst
H
,
Berger
S
,
Erdmann
G
,
Schutz
G
,
Joels
M
. Metaplasticity of amygdalar responses to the stress hormone corticosterone. Proc Natl Acad Sci U S A
107: 14449‐14454, 2010. |
| 101. |
Karst
H
,
Berger
S
,
Turiault
M
,
Tronche
F
,
Schutz
G
,
Joels
M
. Mineralocorticoid receptors are indispensable for nongenomic modulation of hippocampal glutamate transmission by corticosterone. Proc Natl Acad Sci U S A
102: 19204‐19207, 2005. |
| 102. |
Kasai
M
,
Kannan
H
,
Ueta
Y
,
Osaka
T
,
Inenaga
K
,
Yamashita
H
. Effects of iontophoretically applied cortisol on tuberoinfundibular neurons in hypothalamic paraventricular nucleus of anesthetized rats. Neurosci Lett
87: 35‐40, 1988. |
| 103. |
Khan
MS
,
Aden
D
,
Rosner
W
. Human corticosteroid binding globulin is secreted by a hepatoma‐derived cell line. J Steroid Biochem
20: 677‐678, 1984. |
| 104. |
King
DP
,
Zhao
Y
,
Sangoram
AM
,
Wilsbacher
LD
,
Tanaka
M
,
Antoch
MP
,
Steeves
TD
,
Vitaterna
MH
,
Kornhauser
JM
,
Lowrey
PL
,
Turek
FW
,
Takahashi
JS
. Positional cloning of the mouse circadian clock gene. Cell
89: 641‐653, 1997. |
| 105. |
Kino
T
,
Nordeen
SK
,
Chrousos
GP
. Conditional modulation of glucocorticoid receptor activities by CREB‐binding protein (CBP) and p300. J Steroid Biochem Mol Biol
70: 15‐25, 1999. |
| 106. |
Kitchener
P
,
Di Blasi
F
,
Borrelli
E
,
Piazza
PV
. Differences between brain structures in nuclear translocation and DNA binding of the glucocorticoid receptor during stress and the circadian cycle. Eur J Neurosci
19: 1837‐1846, 2004. |
| 107. |
Kow
LM
,
Pfaff
DW
. Vasopressin excites ventromedial hypothalamic glucose‐responsive neurons in vitro. Physiol Behav
37: 153‐158, 1986. |
| 108. |
Kruk
MR
,
Halasz
J
,
Meelis
W
,
Haller
J
. Fast positive feedback between the adrenocortical stress response and a brain mechanism involved in aggressive behavior. Behav Neurosci
118: 1062‐1070, 2004. |
| 109. |
Lamia
KA
,
Storch
KF
,
Weitz
CJ
. Physiological significance of a peripheral tissue circadian clock. Proc Natl Acad Sci U S A
105: 15172‐15177, 2008. |
| 110. | Le
Martelot
G
,
Claudel
T
,
Gatfield
D
,
Schaad
O
,
Kornmann
B
,
Lo Sasso
G
,
Moschetta
A
,
Schibler
U
. REV‐ERBalpha participates in circadian SREBP signaling and bile acid homeostasis. PLoS Biol
7: e1000181, 2009. |
| 111. |
Leliavski
A
,
Shostak
A
,
Husse
J
,
Oster
H
. Impaired glucocorticoid production and response to stress in arntl‐deficient male mice. Endocrinology
155: 133‐142, 2014. |
| 112. |
Levi
F
,
Schibler
U
. Circadian rhythms: Mechanisms and therapeutic implications. Annu Rev Pharmacol Toxicol
47: 593‐628, 2007. |
| 113. |
Lewis
JG
,
Bagley
CJ
,
Elder
PA
,
Bachmann
AW
,
Torpy
DJ
. Plasma free cortisol fraction reflects levels of functioning corticosteroid‐binding globulin. Clin Chim Acta
359: 189‐194, 2005. |
| 114. |
Lewis
JG
,
Mopert
B
,
Shand
BI
,
Doogue
MP
,
Soule
SG
,
Frampton
CM
,
Elder
PA
. Plasma variation of corticosteroid‐binding globulin and sex hormone‐binding globulin. Horm Metab Res
38: 241‐245, 2006. |
| 115. |
Li
TZ
,
Wang
CA
,
Chen
YZ
. [The rapid effect of the iontophoretically applied cortisol on unit activity of neurons in three brain areas in rats]. Sheng li xue bao : [Acta physiologica Sinica]
43: 280‐285, 1991. |
| 116. |
Lightman
SL
,
Conway‐Campbell
BL
. The crucial role of pulsatile activity of the HPA axis for continuous dynamic equilibration. Nat Rev Neurosci
11: 710‐718, 2010. |
| 117. |
Lightman
SL
,
Windle
RJ
,
Julian
MD
,
Harbuz
MS
,
Shanks
N
,
Wood
SA
,
Kershaw
YM
,
Ingram
CD
. Significance of pulsatility in the HPA axis. Novartis Found Symp
227: 244‐257; discussion 257‐260, 2000. |
| 118. |
Lin
D
,
Sugawara
T
,
Strauss
JF, III
,
Clark
BJ
,
Stocco
DM
,
Saenger
P
,
Rogol
A
,
Miller
WL
. Role of steroidogenic acute regulatory protein in adrenal and gonadal steroidogenesis. Science
267: 1828‐1831, 1995. |
| 119. |
Liu
Y
,
Smith
LI
,
Huang
V
,
Poon
V
,
Coello
A
,
Olah
M
,
Spiga
F
,
Lightman
SL
,
Aguilera
G
. Transcriptional regulation of episodic glucocorticoid secretion. Mol Cell Endocrinol
371: 62‐70, 2013. |
| 120. |
Loh
DH
,
Abad
C
,
Colwell
CS
,
Waschek
JA
. Vasoactive intestinal peptide is critical for circadian regulation of glucocorticoids. Neuroendocrinology
88: 246‐255, 2008. |
| 121. |
Macfarlane
DP
,
Forbes
S
,
Walker
BR
. Glucocorticoids and fatty acid metabolism in humans: Fuelling fat redistribution in the metabolic syndrome. J Endocrinol
197: 189‐204, 2008. |
| 122. |
Mahmoud
SN
,
Scaccianoce
S
,
Scraggs
PR
,
Nicholson
SA
,
Gillham
B
,
Jones
MT
. Characteristics of corticosteroid inhibition of adrenocorticotrophin release from the anterior pituitary gland of the rat. J Endocrinol
102: 33‐42, 1984. |
| 123. |
Mangelsdorf
DJ
,
Thummel
C
,
Beato
M
,
Herrlich
P
,
Schutz
G
,
Umesono
K
,
Blumberg
B
,
Kastner
P
,
Mark
M
,
Chambon
P
,
Evans
RM
. The nuclear receptor superfamily: The second decade. Cell
83: 835‐839, 1995. |
| 124. |
Marcheva
B
,
Ramsey
KM
,
Buhr
ED
,
Kobayashi
Y
,
Su
H
,
Ko
CH
,
Ivanova
G
,
Omura
C
,
Mo
S
,
Vitaterna
MH
,
Lopez
JP
,
Philipson
LH
,
Bradfield
CA
,
Crosby
SD
,
JeBailey
L
,
Wang
X
,
Takahashi
JS
,
Bass
J
. Disruption of the clock components CLOCK and BMAL1 leads to hypoinsulinaemia and diabetes. Nature
466: 627‐631, 2010. |
| 125. |
Mataradze
GD
,
Kurabekova
RM
,
Rozen
VB
. The role of sex steroids in the formation of sex‐differentiated concentrations of corticosteroid‐binding globulin in rats. J Endocrinol
132: 235‐240, 1992. |
| 126. |
Maywood
ES
,
O'Neill
JS
,
Chesham
JE
,
Hastings
MH
. Minireview: The circadian clockwork of the suprachiasmatic nuclei-analysis of a cellular oscillator that drives endocrine rhythms. Endocrinology
148: 5624‐5634, 2007. |
| 127. |
McDearmon
EL
,
Patel
KN
,
Ko
CH
,
Walisser
JA
,
Schook
AC
,
Chong
JL
,
Wilsbacher
LD
,
Song
EJ
,
Hong
HK
,
Bradfield
CA
,
Takahashi
JS
. Dissecting the functions of the mammalian clock protein BMAL1 by tissue‐specific rescue in mice. Science
314: 1304‐1308, 2006. |
| 128. |
McEwen
BS
. Physiology and neurobiology of stress and adaptation: Central role of the brain. Physiol Rev
87: 873‐904, 2007. |
| 129. |
Meijer
OC
,
Steenbergen
PJ
,
De Kloet
ER
. Differential expression and regional distribution of steroid receptor coactivators SRC‐1 and SRC‐2 in brain and pituitary. Endocrinology
141: 2192‐2199, 2000. |
| 130. |
Merriam
GR
,
Wachter
KW
. Algorithms for the study of episodic hormone secretion. Am J Physiol
243: E310‐E318, 1982. |
| 131. |
Mershon
JL
,
Sehlhorst
CS
,
Rebar
RW
,
Liu
JH
. Evidence of a corticotropin‐releasing hormone pulse generator in the macaque hypothalamus. Endocrinology
130: 2991‐2996, 1992. |
| 132. |
Metherell
LA
,
Chapple
JP
,
Cooray
S
,
David
A
,
Becker
C
,
Ruschendorf
F
,
Naville
D
,
Begeot
M
,
Khoo
B
,
Nurnberg
P
,
Huebner
A
,
Cheetham
ME
,
Clark
AJ
. Mutations in MRAP, encoding a new interacting partner of the ACTH receptor, cause familial glucocorticoid deficiency type 2. Nat Genet
37: 166‐170, 2005. |
| 133. |
Mikami
K
,
Strott
CA
. Cyclic AMP‐dependent protein kinase activity and protein phosphorylation in zones of the adrenal cortex. Biochem Biophys Res Commun
138: 895‐901, 1986. |
| 134. |
Miller
WL
,
Strauss
JF, 3rd
. Molecular pathology and mechanism of action of the steroidogenic acute regulatory protein, StAR. J Steroid Biochem Mol Biol
69: 131‐141, 1999. |
| 135. |
Mohawk
JA
,
Takahashi
JS
. Cell autonomy and synchrony of suprachiasmatic nucleus circadian oscillators. Trends Neurosci
34: 349‐358, 2011. |
| 136. |
Moore
RY
,
Lenn
NJ
. A retinohypothalamic projection in the rat. J Comp Neurol
146: 1‐14, 1972. |
| 137. |
Mountjoy
KG
,
Mortrud
MT
,
Low
MJ
,
Simerly
RB
,
Cone
RD
. Localization of the melanocortin‐4 receptor (MC4‐R) in neuroendocrine and autonomic control circuits in the brain. Mol Endocrinol
8: 1298‐1308, 1994. |
| 138. |
Munck
A
,
Guyre
PM
,
Holbrook
NJ
. Physiological functions of glucocorticoids in stress and their relation to pharmacological actions. Endoc Rev
5: 25‐44, 1984. |
| 139. |
Nagoshi
E
,
Saini
C
,
Bauer
C
,
Laroche
T
,
Naef
F
,
Schibler
U
. Circadian gene expression in individual fibroblasts: Cell‐autonomous and self‐sustained oscillators pass time to daughter cells. Cell
119: 693‐705, 2004. |
| 140. |
Nishi
M
,
Kawata
M
. Brain corticosteroid receptor dynamics and trafficking: Implications from live cell imaging. Neuroscientist
12: 119‐133, 2006. |
| 141. |
Orchinik
M
,
Murray
TF
,
Franklin
PH
,
Moore
FL
. Guanyl nucleotides modulate binding to steroid receptors in neuronal membranes. Proc Natl Acad Sci U S A
89: 3830‐3834, 1992. |
| 142. |
Orchinik
M
,
Murray
TF
,
Moore
FL
. A corticosteroid receptor in neuronal membranes. Science
252: 1848‐1851, 1991. |
| 143. |
Oster
H
,
Damerow
S
,
Hut
RA
,
Eichele
G
. Transcriptional profiling in the adrenal gland reveals circadian regulation of hormone biosynthesis genes and nucleosome assembly genes. J Biol Rhythms
21: 350‐361, 2006. |
| 144. |
Oster
H
,
Damerow
S
,
Kiessling
S
,
Jakubcakova
V
,
Abraham
D
,
Tian
J
,
Hoffmann
MW
,
Eichele
G
. The circadian rhythm of glucocorticoids is regulated by a gating mechanism residing in the adrenal cortical clock. Cell Metab
4: 163‐173, 2006. |
| 145. |
Park
SY
,
Walker
JJ
,
Johnson
NW
,
Zhao
Z
,
Lightman
SL
,
Spiga
F
. Constant light disrupts the circadian rhythm of steroidogenic proteins in the rat adrenal gland. Mol Cell Endocrinol
371: 114‐123, 2013. |
| 146. |
Pfaff
DW
,
Silva
MT
,
Weiss
JM
. Telemetered recording of hormone effects on hippocampal neurons. Science
172: 394‐395, 1971. |
| 147. |
Picard
D
. Intracellular dynamics of the Hsp90 co‐chaperone p23 is dictated by Hsp90. Exp Cell Res
312: 198‐204, 2006. |
| 148. |
Pon
LA
,
Hartigan
JA
,
Orme‐Johnson
NR
. Acute ACTH regulation of adrenal corticosteroid biosynthesis. Rapid accumulation of a phosphoprotein. J Biol Chem
261: 13309‐13316, 1986. |
| 149. |
Pratt
WB
,
Galigniana
MD
,
Harrell
JM
,
DeFranco
DB
. Role of hsp90 and the hsp90‐binding immunophilins in signalling protein movement. Cell Signal
16: 857‐872, 2004. |
| 150. |
Pratt
WB
,
Morishima
Y
,
Osawa
Y
. The Hsp90 chaperone machinery regulates signaling by modulating ligand binding clefts. J Biol Chem
283: 22885‐22889, 2008. |
| 151. |
Qian
X
,
Droste
SK
,
Gutierrez‐Mecinas
M
,
Collins
A
,
Kersante
F
,
Reul
JM
,
Linthorst
AC
. A rapid release of corticosteroid‐binding globulin from the liver restrains the glucocorticoid hormone response to acute stress. Endocrinology
152: 3738‐3748, 2011. |
| 152. |
Qian
X
,
Droste
SK
,
Lightman
SL
,
Reul
JM
,
Linthorst
AC
. Circadian and ultradian rhythms of free glucocorticoid hormone are highly synchronized between the blood, the subcutaneous tissue, and the brain. Endocrinology
153: 4346‐4353, 2012. |
| 153. |
Ralph
MR
,
Foster
RG
,
Davis
FC
,
Menaker
M
. Transplanted suprachiasmatic nucleus determines circadian period. Science
247: 975‐978, 1990. |
| 154. |
Rankin
J
,
Walker
JJ
,
Windle
R
,
Lightman
SL
,
Terry
JR
. Characterizing dynamic interactions between ultradian glucocorticoid rhythmicity and acute stress using the phase response curve. PloS one
7: e30978, 2012. |
| 155. |
Reppert
SM
,
Weaver
DR
. Coordination of circadian timing in mammals. Nature
418: 935‐941, 2002. |
| 156. |
Reul
JM
,
de Kloet
ER
. Two receptor systems for corticosterone in rat brain: Microdistribution and differential occupation. Endocrinology
117: 2505‐2511, 1985. |
| 157. |
Reul
JM
,
de Kloet
ER
. Anatomical resolution of two types of corticosterone receptor sites in rat brain with in vitro autoradiography and computerized image analysis. J Steroid Biochem
24: 269‐272, 1986. |
| 158. |
Reul
JM
,
de Kloet
ER
,
van Sluijs
FJ
,
Rijnberk
A
,
Rothuizen
J
. Binding characteristics of mineralocorticoid and glucocorticoid receptors in dog brain and pituitary. Endocrinology
127: 907‐915, 1990. |
| 159. |
Rivier
C
,
Vale
W
. Interaction of corticotropin‐releasing factor and arginine vasopressin on adrenocorticotropin secretion in vivo. Endocrinology
113: 939‐942, 1983. |
| 160. |
Rose
JD
,
Moore
FL
,
Orchinik
M
. Rapid neurophysiological effects of corticosterone on medullary neurons: Relationship to stress‐induced suppression of courtship clasping in an amphibian. Neuroendocrinology
57: 815‐824, 1993. |
| 161. |
Rotsztejn
W
,
Lalonde
J
,
Normand
M
,
Fortier
C
. Feedback inhibition of adrenocorticotropin release by corticosterone infusions in the adrenalectomized rat. Can J Physiol Pharmacol
53: 475‐478, 1975. |
| 162. |
Rotsztejn
WH
,
Normand
M
,
Lalonde
J
,
Fortier
C
. Relationship between ACTH release and corticosterone binding by the receptor sites of the adenohypophysis and dorsal hippocampus following infusion of corticosterone at a constant rate in the adrenalectomized rat. Endocrinology
97: 223‐230, 1975. |
| 163. |
Russell
GM
,
Henley
DE
,
Leendertz
J
,
Douthwaite
JA
,
Wood
SA
,
Stevens
A
,
Woltersdorf
WW
,
Peeters
BW
,
Ruigt
GS
,
White
A
,
Veldhuis
JD
,
Lightman
SL
. Rapid glucocorticoid receptor‐mediated inhibition of hypothalamic‐pituitary‐adrenal ultradian activity in healthy males. J Neurosci
30: 6106‐6115, 2010. |
| 164. |
Saphier
D
,
Feldman
S
. Iontophoretic application of glucocorticoids inhibits identified neurones in the rat paraventricular nucleus. Brain Res
453: 183‐190, 1988. |
| 165. |
Sarabdjitsingh
RA
,
Conway‐Campbell
BL
,
Leggett
JD
,
Waite
EJ
,
Meijer
OC
,
de Kloet
ER
,
Lightman
SL
. Stress responsiveness varies over the ultradian glucocorticoid cycle in a brain‐region‐specific manner. Endocrinology
151: 5369‐5379, 2010. |
| 166. |
Sarabdjitsingh
RA
,
Isenia
S
,
Polman
A
,
Mijalkovic
J
,
Lachize
S
,
Datson
N
,
de Kloet
ER
,
Meijer
OC
. Disrupted corticosterone pulsatile patterns attenuate responsiveness to glucocorticoid signaling in rat brain. Endocrinology
151: 1177‐1186, 2010. |
| 167. |
Sawchenko
PE
,
Swanson
LW
,
Vale
WW
. Co‐expression of corticotropin‐releasing factor and vasopressin immunoreactivity in parvocellular neurosecretory neurons of the adrenalectomized rat. Proc Natl Acad Sci U S A
81: 1883‐1887, 1984. |
| 168. |
Schoneveld
OJ
,
Gaemers
IC
,
Lamers
WH
. Mechanisms of glucocorticoid signalling. Biochim Biophys Acta
1680: 114‐128, 2004. |
| 169. |
Seale
JV
,
Wood
SA
,
Atkinson
HC
,
Bate
E
,
Lightman
SL
,
Ingram
CD
,
Jessop
DS
,
Harbuz
MS
. Gonadectomy reverses the sexually diergic patterns of circadian and stress‐induced hypothalamic‐pituitary‐adrenal axis activity in male and female rats. J Neuroendocrinol
16: 516‐524, 2004. |
| 170. |
Seale
JV
,
Wood
SA
,
Atkinson
HC
,
Harbuz
MS
,
Lightman
SL
. Gonadal steroid replacement reverses gonadectomy‐induced changes in the corticosterone pulse profile and stress‐induced hypothalamic‐pituitary‐adrenal axis activity of male and female rats. J Neuroendocrinol
16: 989‐998, 2004. |
| 171. |
Seale
JV
,
Wood
SA
,
Atkinson
HC
,
Harbuz
MS
,
Lightman
SL
. Postnatal masculinization alters the HPA axis phenotype in the adult female rat. J Physiol
563: 265‐274, 2005. |
| 172. |
Seale
JV
,
Wood
SA
,
Atkinson
HC
,
Lightman
SL
,
Harbuz
MS
. Organizational role for testosterone and estrogen on adult hypothalamic‐pituitary‐adrenal axis activity in the male rat. Endocrinology
146: 1973‐1982, 2005. |
| 173. |
Shanks
N
,
Lightman
SL
. The maternal‐neonatal neuro‐immune interface: Are there long‐term implications for inflammatory or stress‐related disease? J Clin Invest
108: 1567‐1573, 2001. |
| 174. |
Shanks
N
,
Windle
RJ
,
Perks
PA
,
Harbuz
MS
,
Jessop
DS
,
Ingram
CD
,
Lightman
SL
. Early‐life exposure to endotoxin alters hypothalamic‐pituitary‐adrenal function and predisposition to inflammation. Proc Natl Acad Sci U S A
97: 5645‐5650, 2000. |
| 175. |
Son
GH
,
Chung
S
,
Choe
HK
,
Kim
HD
,
Baik
SM
,
Lee
H
,
Lee
HW
,
Choi
S
,
Sun
W
,
Kim
H
,
Cho
S
,
Lee
KH
,
Kim
K
. Adrenal peripheral clock controls the autonomous circadian rhythm of glucocorticoid by causing rhythmic steroid production. Proc Natl Acad Sci U S A
105: 20970‐20975, 2008. |
| 176. |
Song
KH
,
Park
JI
,
Lee
MO
,
Soh
J
,
Lee
K
,
Choi
HS
. LH induces orphan nuclear receptor Nur77 gene expression in testicular Leydig cells. Endocrinology
142: 5116‐5123, 2001. |
| 177. |
Spencer
RL
,
Miller
AH
,
Moday
H
,
Stein
M
,
McEwen
BS
. Diurnal differences in basal and acute stress levels of type I and type II adrenal steroid receptor activation in neural and immune tissues. Endocrinology
133: 1941‐1950, 1993. |
| 178. |
Spiga
F
,
Harrison
LR
,
Wood
SA
,
MacSweeney
CP
,
Thomson
FJ
,
Craighead
M
,
Grassie
M
,
Lightman
SL
. Effect of the glucocorticoid receptor antagonist Org 34850 on fast and delayed feedback of corticosterone release. J Endocrinol
196: 323‐330, 2008. |
| 179. |
Spiga
F
,
Liu
Y
,
Aguilera
G
,
Lightman
SL
. Temporal effect of adrenocorticotrophic hormone on adrenal glucocorticoid steroidogenesis: Involvement of the transducer of regulated cyclic AMP‐response element‐binding protein activity. J Neuroendocrinol
23: 136‐142, 2011. |
| 180. |
Spiga
F
,
Waite
EJ
,
Liu
Y
,
Kershaw
YM
,
Aguilera
G
,
Lightman
SL
. ACTH‐Dependent Ultradian Rhythm of Corticosterone Secretion. Endocrinology
152: 1448‐1457, 2011. |
| 181. |
Stavreva
DA
,
Wiench
M
,
John
S
,
Conway‐Campbell
BL
,
McKenna
MA
,
Pooley
JR
,
Johnson
TA
,
Voss
TC
,
Lightman
SL
,
Hager
GL
. Ultradian hormone stimulation induces glucocorticoid receptor‐mediated pulses of gene transcription. Nat Cell Biol
11: 1093‐1102, 2009. |
| 182. |
Stephan
FK
,
Zucker
I
. Circadian rhythms in drinking behavior and locomotor activity of rats are eliminated by hypothalamic lesions. Proc Natl Acad Sci U S A
69: 1583‐1586, 1972. |
| 183. |
Sternberg
EM
,
Hill
JM
,
Chrousos
GP
,
Kamilaris
T
,
Listwak
SJ
,
Gold
PW
,
Wilder
RL
. Inflammatory mediator‐induced hypothalamic‐pituitary‐adrenal axis activation is defective in streptococcal cell wall arthritis‐susceptible Lewis rats. Proc Natl Acad Sci U S A
86: 2374‐2378, 1989. |
| 184. |
Sternberg
EM
,
Young
WS, 3rd
,
Bernardini
R
,
Calogero
AE
,
Chrousos
GP
,
Gold
PW
,
Wilder
RL
. A central nervous system defect in biosynthesis of corticotropin‐releasing hormone is associated with susceptibility to streptococcal cell wall‐induced arthritis in Lewis rats. Proc Natl Acad Sci U S A
86: 4771‐4775, 1989. |
| 185. |
Stocco
DM
,
Clark
BJ
. Regulation of the acute production of steroids in steroidogenic cells. Endoc Rev
17: 221‐244, 1996a. |
| 186. |
Stocco
DM
,
Clark
BJ
. Role of the steroidogenic acute regulatory protein (StAR) in steroidogenesis. Biochem Pharmacol
51: 197‐205, 1996b. |
| 187. |
Stokkan
KA
,
Yamazaki
S
,
Tei
H
,
Sakaki
Y
,
Menaker
M
. Entrainment of the circadian clock in the liver by feeding. Science
291: 490‐493, 2001. |
| 188. |
Stratmann
M
,
Schibler
U
. Properties, entrainment, and physiological functions of mammalian peripheral oscillators. J Biol Rhythms
21: 494‐506, 2006. |
| 189. |
Sugawara
T
,
Holt
JA
,
Kiriakidou
M
,
Strauss
JF, 3rd
. Steroidogenic factor 1‐dependent promoter activity of the human steroidogenic acute regulatory protein (StAR) gene. Biochemistry
35: 9052‐9059, 1996. |
| 190. |
Takemori
H
,
Kanematsu
M
,
Kajimura
J
,
Hatano
O
,
Katoh
Y
,
Lin
XZ
,
Min
L
,
Yamazaki
T
,
Doi
J
,
Okamoto
M
. Dephosphorylation of TORC initiates expression of the StAR gene. Mol Cell Endocrinol
265‐266: 196‐204, 2007. |
| 191. |
Takemori
H
,
Okamoto
M
. Regulation of CREB‐mediated gene expression by salt inducible kinase. J Steroid Biochem Mol Biol
108: 287‐291, 2008. |
| 192. |
Tapp
WN
,
Holaday
JW
,
Natelson
BH
. Ultradian glucocorticoid rhythms in monkeys and rats continue during stress. Am J Physiol
247: R866‐R871, 1984. |
| 193. |
Tasker
JG
,
Herman
JP
. Mechanisms of rapid glucocorticoid feedback inhibition of the hypothalamic‐pituitary‐adrenal axis. Stress
14: 398‐406, 2011. |
| 194. |
Tousson
E
,
Meissl
H
. Suprachiasmatic nuclei grafts restore the circadian rhythm in the paraventricular nucleus of the hypothalamus. J Neurosci
24: 2983‐2988, 2004. |
| 195. |
Ulrich‐Lai
YM
,
Arnhold
MM
,
Engeland
WC
. Adrenal splanchnic innervation contributes to the diurnal rhythm of plasma corticosterone in rats by modulating adrenal sensitivity to ACTH. Am J Physiol Regul Integr Comp Physiol
290: R1128‐R1135, 2006. |
| 196. |
Ulrich‐Lai
YM
,
Herman
JP
. Neural regulation of endocrine and autonomic stress responses. Nat Rev Neurosci
10: 397‐409, 2009. |
| 197. |
Vale
W
,
Spiess
J
,
Rivier
C
,
Rivier
J
. Characterization of a 41‐residue ovine hypothalamic peptide that stimulates secretion of corticotropin and beta‐endorphin. Science
213: 1394‐1397, 1981. |
| 198. |
Van Eekelen
JA
,
De Kloet
ER
. Co‐localization of brain corticosteroid receptors in the rat hippocampus. Prog Histochem Cytochem
26: 250‐258, 1992. |
| 199. |
Vrang
N
,
Larsen
PJ
,
Mikkelsen
JD
. Direct projection from the suprachiasmatic nucleus to hypophysiotrophic corticotropin‐releasing factor immunoreactive cells in the paraventricular nucleus of the hypothalamus demonstrated by means of Phaseolus vulgaris‐leucoagglutinin tract tracing. Brain Res
684: 61‐69, 1995. |
| 200. |
Waite
EJ
,
McKenna
M
,
Kershaw
Y
,
Walker
JJ
,
Cho
K
,
Piggins
HD
,
Lightman
SL
. Ultradian corticosterone secretion is maintained in the absence of circadian cues. Eur J Neurosci
36: 3142‐3150, 2012. |
| 201. |
Walker
JJ
,
Spiga
F
,
Waite
E
,
Zhao
Z
,
Kershaw
Y
,
Terry
JR
,
Lightman
SL
. The origin of glucocorticoid hormone oscillations. PLoS Biol
10: e1001341, 2012. |
| 202. |
Walker
JJ
,
Terry
JR
,
Lightman
SL
. Origin of ultradian pulsatility in the hypothalamic‐pituitary‐adrenal axis. Proc Biol Sci R Soc
277: 1627‐1633, 2010. |
| 203. |
Wallberg
AE
,
Flinn
EM
,
Gustafsson
JA
,
Wright
AP
. Recruitment of chromatin remodelling factors during gene activation via the glucocorticoid receptor N‐terminal domain. Biochem Soc Trans
28: 410‐414, 2000. |
| 204. |
Watts
AG
,
Swanson
LW
. Efferent projections of the suprachiasmatic nucleus: II. Studies using retrograde transport of fluorescent dyes and simultaneous peptide immunohistochemistry in the rat. J Comp Neurol
258: 230‐252, 1987. |
| 205. |
Watts
AG
,
Swanson
LW
,
Sanchez‐Watts
G
. Efferent projections of the suprachiasmatic nucleus: I. Studies using anterograde transport of Phaseolus vulgaris leucoagglutinin in the rat. J Comp Neurol
258: 204‐229, 1987. |
| 206. |
Watts
AG
,
Tanimura
S
,
Sanchez‐Watts
G
. Corticotropin‐releasing hormone and arginine vasopressin gene transcription in the hypothalamic paraventricular nucleus of unstressed rats: Daily rhythms and their interactions with corticosterone. Endocrinology
145: 529‐540, 2004. |
| 207. |
Weitzman
ED
,
Fukushima
D
,
Nogeire
C
,
Roffwarg
H
,
Gallagher
TF
,
Hellman
L
. Twenty‐four hour pattern of the episodic secretion of cortisol in normal subjects. J Clin Endocrinol Metab
33: 14‐22, 1971. |
| 208. |
Welsh
DK
,
Yoo
SH
,
Liu
AC
,
Takahashi
JS
,
Kay
SA
. Bioluminescence imaging of individual fibroblasts reveals persistent, independently phased circadian rhythms of clock gene expression. Curr Biol
14: 2289‐2295, 2004. |
| 209. |
Widmaier
EP
,
Dallman
MF
. The effects of corticotropin‐releasing factor on adrenocorticotropin secretion from perifused pituitaries in vitro: Rapid inhibition by glucocorticoids. Endocrinology
115: 2368‐2374, 1984. |
| 210. |
Wilkinson
CW
,
Shinsako
J
,
Dallman
MF
. Return of pituitary‐adrenal function after adrenal enucleation or transplantation: Diurnal rhythms and responses to ether. Endocrinology
109: 162‐169, 1981. |
| 211. |
Windle
RJ
,
Wood
SA
,
Kershaw
YM
,
Lightman
SL
,
Ingram
CD
. Adaptive changes in basal and stress‐induced HPA activity in lactating and post‐lactating female rats. Endocrinology
154: 749‐761, 2013. |
| 212. |
Windle
RJ
,
Wood
SA
,
Kershaw
YM
,
Lightman
SL
,
Ingram
CD
,
Harbuz
MS
. Increased corticosterone pulse frequency during adjuvant‐induced arthritis and its relationship to alterations in stress responsiveness. J Neuroendocrinol
13: 905‐911, 2001. |
| 213. |
Windle
RJ
,
Wood
SA
,
Lightman
SL
,
Ingram
CD
. The pulsatile characteristics of hypothalamo‐pituitary‐adrenal activity in female Lewis and Fischer 344 rats and its relationship to differential stress responses. Endocrinology
139: 4044‐4052, 1998. |
| 214. |
Windle
RJ
,
Wood
SA
,
Shanks
N
,
Lightman
SL
,
Ingram
CD
. Ultradian rhythm of basal corticosterone release in the female rat: Dynamic interaction with the response to acute stress. Endocrinology
139: 443‐450, 1998. |
| 215. |
Wochnik
GM
,
Ruegg
J
,
Abel
GA
,
Schmidt
U
,
Holsboer
F
,
Rein
T
. FK506‐binding proteins 51 and 52 differentially regulate dynein interaction and nuclear translocation of the glucocorticoid receptor in mammalian cells. J Biol Chem
280: 4609‐4616, 2005. |
| 216. |
Yamazaki
S
,
Numano
R
,
Abe
M
,
Hida
A
,
Takahashi
R
,
Ueda
M
,
Block
GD
,
Sakaki
Y
,
Menaker
M
,
Tei
H
. Resetting central and peripheral circadian oscillators in transgenic rats. Science
288: 682‐685, 2000. |
| 217. |
Zazopoulos
E
,
Lalli
E
,
Stocco
DM
,
Sassone‐Corsi
P
. DNA binding and transcriptional repression by DAX‐1 blocks steroidogenesis. Nature
390: 311‐315, 1997. |